Quantum: Einstein, Bohr and the Great Debate About the Nature of Reality - Manjit Kumar (2009)
Part I. THE QUANTUM
Chapter 6. THE PRINCE OF DUALITY
'Science is an old lady who does not fear mature men', his father had once said.1 But he, like his elder brother, had been seduced by science. Prince Louis Victor Pierre Raymond de Broglie, a member of one of France's leading aristocratic families, had been expected to follow in the footsteps of his illustrious forebears. The de Broglie family, originally from Piedmont, had served French kings as soldiers, statesmen and diplomats with high distinction since the middle of the seventeenth century. In recognition of the service he had rendered, an ancestor was given the hereditary title of Duc in 1742 by Louis XV. The Duke's son, Victor-François, inflicted a crushing defeat on an enemy of the Holy Roman Empire and a grateful Emperor rewarded him with the title of Prinz. Henceforth, all of his descendants would be either a prince or a princess. So it was that the young scientist would one day be both a German prince and a French duke.2 It is an unlikely family history for the man who made a fundamental contribution to quantum physics, which Einstein described as 'the first feeble ray of light on this worst of our physics enigmas'.3
![]()
The youngest of the four surviving children, Louis was born in Dieppe on 15 August 1892. In keeping with their elevated position in society, the de Broglies were educated at the ancestral home by private tutors. While other boys might have been able to recite the names of the great steam engines of the day, Louis could recite the names of all the ministers of the Third Republic. To the amusement of the family, he began giving speeches based upon the political coverage in the newspapers. With a grandfather who had been prime minister, before long 'a great future as a statesman was predicted for Louis', recalled his sister Pauline.4 It might have been the case had his father not died, in 1906, when he was fourteen.
His elder brother, Maurice, at 31, was now the head of the family. As tradition demanded, Maurice had pursued a military career but had chosen the navy rather than the army. At naval college he excelled at science. As a promising young officer he found a navy in a period of transition as it prepared for the twentieth century. Given his scientific interests, it was only a matter of time before Maurice became involved in attempts at establishing a reliable ship-to-ship wireless communication system. In 1902 he wrote his first paper on 'radioelectric waves' and, despite the opposition of his father, it strengthened his determination to leave the navy and devote himself to scientific research. In 1904, after nine years in the service, he quit the navy. Two years later his father was dead and he had to shoulder new responsibilities as the sixth Duc.
On Maurice's advice, Louis was sent to school. 'Having experienced myself the inconvenience of a pressure exercised on the studies of a young man I refrained from imparting a rigid direction to the studies of my brother, although at times his vacillations gave me some concerns', he wrote almost half a century later.5 Louis did well in French, history, physics and philosophy. In mathematics and chemistry he was indifferent. After three years Louis graduated in 1909 at the age of seventeen, with both the baccalauréat of philosophy and that of mathematics. A year earlier Maurice had acquired his PhD under Paul Langevin at the Collège de France and set up a laboratory in his Parisian mansion on the rue Châteaubriand. Rather than seek employment in a university, the creation of a private laboratory in which to pursue his new vocation helped soften the disappointment of some of the family at a de Broglie abandoning military service for science.
Unlike Maurice, Louis at the time was set for a more traditional career as he studied medieval history at the University of Paris. However, the twenty-year-old prince soon discovered that the critical study of texts, sources and documents of the past held little interest for him. Maurice said later that his brother was 'not far from losing faith in himself'.6 Part of the problem was a burgeoning interest in physics fostered by time spent with Maurice in the laboratory. The enthusiasm of his elder brother about his research on X-rays had proved contagious. However, Louis was consumed by doubts about his abilities that were aggravated by failing a physics exam. Was he, Louis wondered, destined to be a failure? 'Gone the gaiety and high spirits of his adolescence! The brilliant chatter of his childhood has been muted by the depth of his reflections', was how Maurice remembered the introvert he hardly recognised.7 Louis would become, according to his brother, 'an austere and fairly untamed scholar', who did not like leaving his own home.8
The first time Louis travelled abroad it was to Brussels in October 1911.9 He was nineteen. In the years since he left the navy, Maurice had become a much-respected scientist specialising in X-ray physics. When the invitation arrived to be one of the two scientific secretaries entrusted with the smooth running of the first Solvay conference, he readily accepted. Even though it was an administrator's role, the chance to discuss the quantum with the likes of Planck, Einstein and Lorentz was just too tempting to forgo. The French would be well represented. Curie, Poincaré, Perrin, and his former supervisor Langevin would all be there.
Staying at the Hotel Metropole with all the delegates, Louis kept his distance. It was only after they returned and Maurice recounted the discussions about the quantum that took place in the small room on the first floor that Louis began taking an even greater interest in the new physics. When the proceedings of the conference were published, Louis read them and resolved to become a physicist. By then he had already swapped history books for those of physics, and in 1913 he obtained his Licence és Science, the equivalent of a degree. His plans had to wait as a year of military service beckoned. Despite the three Marshals of France that the de Broglies could boast, Louis entered the army as a lowly private in a company of engineers stationed just outside Paris.10 With Maurice's help, he was soon transferred to the Service of Wireless Communication. Any hopes of a quick return to his study of physics evaporated with the outbreak of the First World War. He spent the next four years as a radio engineer stationed underneath the Eiffel Tower.
Discharged in August 1919, he deeply resented having spent six years, from the age of 21 to 27, in uniform. Louis was more determined than ever to continue down his chosen path. He was helped and encouraged by Maurice and spent time in his well-equipped laboratory following the research being done on X-rays and the photoelectric effect. The brothers had long discussions on the interpretation of the experiments being conducted. Maurice reminded Louis of 'the educational value of the experimental sciences' and 'that theoretical constructions of science have no value unless they are supported by facts'.11 He wrote a series of papers on the absorption of X-rays while thinking about the nature of electromagnetic radiation. The brothers accepted that both the wave and particle theories of light were in some sense correct, since neither on its own could explain diffraction and interference and also the photoelectric effect.
In 1922, the year Einstein lectured in Paris at the invitation of Langevin and received a hostile reception for having remained in Berlin throughout the war, de Broglie wrote a paper in which he explicitly adopted 'the hypothesis of quanta of light'. He had already accepted the existence of 'atoms of light' at a time when Compton had yet to make any sort of announcement concerning his experiments. By the time the American published his data and analysis of the scattering of X-rays by electrons and thereby confirmed the reality of Einstein's light-quanta, de Broglie had already learned to live with the strange duality of light. Others, however, were only half-joking when they complained about having to teach the wave theory of light on Mondays, Wednesdays and Fridays, and the particle theory on Tuesdays, Thursdays and Saturdays.
'After long reflection in solitude and meditation,' de Broglie wrote later, 'I suddenly had the idea, during the year 1923, that the discovery made by Einstein in 1905 should be generalized by extending it to all material particles and notably to electrons.'12 De Broglie had dared to ask the simple question: if light waves can behave like particles, can particles such as electrons behave like waves? His answer was yes, as de Broglie discovered that if he assigned to an electron a 'fictitious associated wave' with a frequency v and wavelength ![]() , he could explain the exact location of the orbits in Bohr's quantum atom. An electron could occupy only those orbits that could accommodate a whole number of wavelengths of its 'fictitious associated wave'.
, he could explain the exact location of the orbits in Bohr's quantum atom. An electron could occupy only those orbits that could accommodate a whole number of wavelengths of its 'fictitious associated wave'.
In 1913, to prevent Rutherford's model of the hydrogen atom from collapsing as its orbiting electron radiated energy and spiralled into the nucleus, Bohr had been forced to impose a condition for which he could offer no other justification: an electron in a stationary orbit around the nucleus did not emit radiation. De Broglie's idea of treating electrons as standing waves was a radical departure from thinking about electrons as particles orbiting an atomic nucleus.
Standing waves can easily be generated in strings tethered at both ends, such as those used in violins and guitars. Plucking such a string produces a variety of standing waves with the defining characteristic that they are made up of a whole number of half-wavelengths. The longest standing wave possible is one with a wavelength twice as long as the string. The next standing wave is made up of two such half-wavelength units, giving a wavelength equal to the physical length of the string. The next is a standing wave consisting of three half-wavelengths, and so on up the scale. This whole number sequence of standing waves is the only one that is physically possible, and each has its own energy. Given the relationship between frequency and wavelength, this is equivalent to the fact that a plucked guitar string can vibrate only at certain frequencies beginning with the fundamental tone, the lowest frequency.
De Broglie realised that this 'whole number' condition restricted the possible electron orbits in the Bohr atom to those with a circumference that permitted the formation of standing waves. These electron standing waves were not bound at either end like those on a musical instrument, but
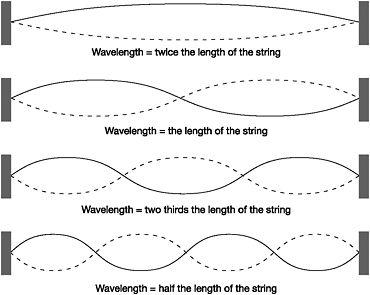
Figure 9: Standing waves of a string tethered at both ends
were formed because a whole number of half-wavelengths could be fitted into the circumference of the orbit. Where there was no exact fit, there could be no standing wave and therefore no stationary orbit.
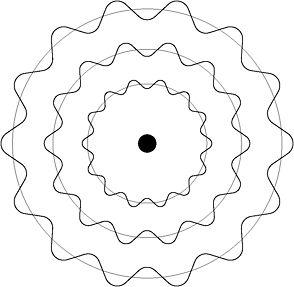
Figure 10: Standing electron waves in the quantum atom
If viewed as a standing wave around the nucleus instead of a particle in orbit, an electron would experience no acceleration and therefore no continual loss of radiation sending it crashing into the nucleus as the atom collapsed. What Bohr had introduced simply to save his quantum atom, found its justification in de Broglie's wave-particle duality. When he did the calculations, de Broglie found that Bohr's principal quantum number, n, labelled only those orbits in which electron standing waves could exist around the nucleus of the hydrogen atom. It was the reason why all other electron orbits were forbidden in the Bohr model.
When de Broglie outlined why all particles should be viewed as having a dual wave-particle character in three short papers in the autumn of 1923, it was not immediately clear what was the nature of the relationship between the billiard ball-like particles and the 'fictitious associated wave'. Was de Broglie suggesting that it was akin to a surfer riding a wave? It was later established that such an interpretation would not work and that electrons, and all other particles, behaved exactly like photons: they are both wave and particle.
De Broglie wrote up his ideas in an expanded form and presented them as his PhD thesis in the spring of 1924. The necessary formalities of acceptance and its reading by the examiners meant that de Broglie did not defend his doctoral dissertation until 25 November. Three of the four examiners were professors at the Sorbonne: Jean Perrin, who had been instrumental in testing Einstein's theory of Brownian motion; Charles Mauguin, a distinguished physicist working on the properties of crystals; and Elie Cartan, a renowned mathematician. The last member of the quartet was the external examiner, Paul Langevin. He alone was well versed in quantum physics and relativity. Before officially submitting his dissertation, de Broglie approached Langevin and asked him to look at his conclusions. Langevin agreed and afterwards told a colleague: 'I am taking with me the little brother's thesis. Looks far-fetched to me.'13
Louis de Broglie's ideas may have been fanciful, but Langevin did not quickly dismiss them. He needed to consult another. Langevin knew that Einstein had publicly stated in 1909 that future research into radiation would reveal a kind of fusion of the particle and wave. Compton's experiments had convinced almost everyone that Einstein had been right about light. It did after all appear to be a particle in collisions with electrons. Now, de Broglie was suggesting the same kind of fusion, wave-particle duality, for all of matter. He even had a formula that linked the wavelength ![]() of the 'particle' to its momentum p,
of the 'particle' to its momentum p, ![]() =h/p where h is Planck's constant. Langevin asked the physicist prince for a second copy of the dissertation and sent it to Einstein. 'He has lifted a corner of the great veil', Einstein wrote back to Langevin.14
=h/p where h is Planck's constant. Langevin asked the physicist prince for a second copy of the dissertation and sent it to Einstein. 'He has lifted a corner of the great veil', Einstein wrote back to Langevin.14
The judgement of Einstein was enough for Langevin and the other examiners. They congratulated de Broglie for 'having pursued with a remarkable mastery an effort that had to be attempted in order to overcome the difficulties in the midst of which the physicists found themselves'.15 Mauguin later admitted that he 'did not believe at the time in the physical reality of the waves associated with grains of matter'.16 All Perrin knew for sure was that de Broglie was 'very intelligent'.17 As for the rest he had no idea. With Einstein's support, aged 32, he was no longer just Prince Louis Victor Pierre Raymond de Broglie, but had earned the right to call himself plain Dr Louis de Broglie.
Having an idea was one thing, but could it be tested? De Broglie had quickly realised in September 1923 that if matter has wave properties, then a beam of electrons should spread out like a beam of light - they should be diffracted. In one of his short papers written that year, de Broglie had predicted that a 'group of electrons that passes through a small aperture should show diffraction effects'.18 He tried, but failed to convince any of the skilled experimentalists working in his brother's private laboratory to put his idea to the test. Busy with other projects, they simply thought the experiments too difficult to perform. Already indebted to his brother Maurice for continually directing his 'attention to the importance and the undeniable accuracy of the dual particulate and wave properties of radiation', Louis did not pursue the matter.19
However Walter Elsasser, a young physicist at Göttingen University, soon pointed out that if de Broglie was right, a simple crystal would diffract a beam of electrons hitting it: since the spacing between adjacent atoms in a crystal would be small enough for an object the size of an electron to reveal its wave character. 'Young man, you are sitting on a gold mine', Einstein told Elsasser when he heard of his proposed experiment.20 It was not a gold mine, but something a bit more precious: a Nobel Prize. But as in any gold rush, one cannot wait too long before getting started. Elsasser did, and two others staked their claims first and grabbed the prize.
Thirty-four-year-old Clinton Davisson of the Western Electric Company in New York, later better known as the Bell Telephone Laboratories, had been investigating the consequences of smashing a beam of electrons into various metal targets when, one day in April 1925, a strange thing happened. A bottle of liquefied air exploded in his laboratory and broke the evacuated tube containing the nickel target that he was using. The air caused the nickel to rust. As a result of cleaning the nickel by heating it, Davisson had accidentally turned the array of tiny nickel crystals into just a few large ones, which caused electron diffraction. When he continued his experiments he soon realised that his results were different. Unaware that he had diffracted electrons, he simply wrote up the data and published it.
'It seems impossible that we will be in Oxford a month from today - doesn't it? We should have a lovely time - Lottie darling - It will be a second honeymoon - and should be sweeter even than the first', Davisson wrote to his wife in July 1926.21 With the children being looked after by relatives back home, the Davissons could enjoy a much-needed break touring England before heading to Oxford and the British Association for the Advancement of Science conference. It was there that Davisson was astonished to learn that some physicists believed that the data from his experiment supported the idea of a French prince. He had not heard of de Broglie or his suggestion that wave-particle duality be extended to encompass all matter. Davisson was not alone.
Few people had read de Broglie's three short papers because they had been published in the French journal Compte Rendu. Fewer still knew of the existence of the doctoral dissertation. On returning to New York, Davisson and a colleague, Lester Germer, immediately set about checking whether electrons really were diffracted. It was January 1927 before they had conclusive evidence that matter was diffracted, it did behave like waves, when Davisson calculated the wavelengths of the diffracted electrons from the new results and found they matched those predicted by de Broglie's theory of wave-particle duality. Davisson later admitted that the original experiments were really 'undertaken as a sort of sideline' in the wake of others that he had been conducting on behalf of his employers, who were defending a lawsuit from a rival company.
Max Knoll and Ernst Ruska quickly utilised the wave nature of the electron with the invention in 1931 of the electron microscope. No particle smaller than approximately half the wavelength of white light can absorb or reflect light waves so as to make the particle visible through an ordinary microscope. However, with wavelengths more than 100,000 times smaller than that of light, electron waves could. The construction of the first commercial electron microscope began in England in 1935.
Meanwhile in Aberdeen, Scotland, the English physicist George Paget Thomson was carrying out his own experiments with electron beams as Davisson and Germer were busy conducting theirs. He too had attended the BAAS conference in Oxford where de Broglie's work had been widely discussed. Thomson, who had a very personal interest in the nature of the electron, immediately began experiments to detect electron diffraction. But instead of crystals, he used specially prepared thin films that gave a diffraction pattern whose features were exactly as de Broglie predicted. Sometimes matter behaves like a wave, smeared over an extended region of space, and at others like a particle, located at a single position in space.
In a remarkable twist of fate, the dual nature of matter was embodied in the Thomson family. George Thomson was awarded the Nobel Prize for physics in 1937, together with Davisson, for discovering that the electron was a wave. His father, Sir J.J. Thomson, had been awarded the Nobel Prize for physics in 1906 for discovering that the electron was a particle.
![]()
Over a quarter of a century, the developments in quantum physics - from Planck's blackbody radiation law to Einstein's quantum of light, from Bohr's quantum atom to de Broglie's wave-particle duality of matter - were the product of an unhappy marriage of quantum concepts and classical physics. It was a union that by 1925 was increasingly under strain. 'The more successes the quantum theory enjoys, the more stupid it looks', Einstein had written as early as May 1912.22 What was needed was a new theory, a new mechanics of the quantum world.
'The discovery of quantum mechanics in the mid-1920s,' said the American Nobel laureate Steven Weinberg, 'was the most profound revolution in physical theory since the birth of modern physics in the seventeenth century.'23Given the pivotal role of young physicists in making the revolution that shaped the modern world, these were the years of knabenphysik - 'boy physics'.