Quantum: Einstein, Bohr and the Great Debate About the Nature of Reality - Manjit Kumar (2009)
Part I. THE QUANTUM
Chapter 3. THE GOLDEN DANE
Manchester, England, Wednesday, 19 June 1912. 'Dear Harald, Perhaps I have found out a little about the structure of atoms,' Niels Bohr wrote to his younger brother.1 'Don't talk about it to anybody,' he warned, 'for otherwise I couldn't write to you so soon.' Silence was essential for Bohr, as he hoped to do what every scientist dreams of: unveiling 'a little bit of reality'. There was still work to be done and he was 'eager to finish it in a hurry, and to do that I have taken off a couple of days from the laboratory (this is also a secret)'. It would take the 26-year-old Dane much longer than he thought to turn his fledgling ideas into a trilogy of papers all entitled 'On the Constitution of Atoms and Molecules'. The first, published in July 1913, was truly revolutionary, as Bohr introduced the quantum directly into the atom.
![]()
It was his mother Ellen's 25th birthday when Niels Henrik David Bohr was born on 7 October 1885 in Copenhagen. She had returned to the comfort of her parents' home for the birth of her second child. Across the wide cobbled street from Christianborg Castle, the seat of the Danish parliament, Ved Stranden 14 was one of the most magnificent residences in the city. A banker and politician, her father was one of the wealthiest men in Denmark. Although the Bohrs did not stay there long, it was to be the first of the grand and elegant homes in which Niels lived throughout his life.
Christian Bohr was the distinguished professor of physiology at Copenhagen University. He had discovered the role of carbon dioxide in the release of oxygen by haemoglobin, and together with his research on respiration it led to nominations for the Nobel Prize for physiology or medicine. From 1886 until his untimely death in 1911, at just 56, the family lived in a spacious apartment in the university's Academy of Surgery.2 Situated in the city's most fashionable street and a ten-minute walk from the local school, it was ideal for the Bohr children: Jenny, two years older than Niels, and Harald, eighteen months younger.3 With three maids and a nanny to look after them, they enjoyed a comfortable and privileged childhood far removed from the squalid and overcrowded conditions in which most of Copenhagen's ever-increasing inhabitants lived.
His father's academic position and his mother's social standing ensured that many of Denmark's leading scientists and scholars, writers and artists were regular visitors to the Bohr home. Three such guests were, like Bohr senior, members of the Royal Danish Academy of Sciences and Letters: the physicist Christian Christiansen, the philosopher Harald Høffding and the linguist Vilhelm Thomsen. After the Academy's weekly meeting, the discussion would continue at the home of one of the quartet. In their teens, whenever their father played host to his fellow Academicians, Niels and Harald were allowed to eavesdrop on the animated debates that took place. It was a rare opportunity to listen to the intellectual concerns of a group of such men as the mood of fin-de-siècle gripped Europe. They left on the boys, as Niels said later, 'some of our earliest and deepest impressions'.4
Bohr the schoolboy excelled at mathematics and science, but had little aptitude for languages. 'In those days,' recalled a friend, 'he was definitely not afraid to use his strength when it came to blows during the break between classes.'5 By the time he enrolled at Copenhagen University, then Denmark's only university, to study physics in 1903, Einstein had spent more than a year at the Patent Office in Bern.6 When he received his Master's degree in 1909, Einstein was extraordinary professor of theoretical physics at the University of Zurich and had received his first nomination for the Nobel Prize. Bohr had also distinguished himself, albeit on a far smaller stage. In 1907, aged 21, he won the Gold Medal of the Royal Danish Academy with a paper on the surface tension of water. It was the reason why his father, who had won the silver medal in 1885, often proudly proclaimed, 'I'm silver but Niels is gold'.7
Bohr struck gold after his father persuaded him to abandon the laboratory for a place in the countryside to finish writing his award-winning paper. Although he submitted it just hours before the deadline, Bohr still found something to add, and handed in a postscript two days later. The need to rework any piece of writing until he was satisfied that it conveyed exactly what he wanted verged on an obsession. A year before he finished his doctoral thesis, Bohr admitted that he had already written 'fourteen more or less divergent rough drafts'.8 Even the simple act of penning a letter became a protracted affair. One day Harald, seeing a letter lying on Niels' desk, offered to post it, only to be told: 'Oh no, that is just one of the first drafts for a rough copy.'9
All their lives, the brothers remained the closest of friends. Apart from mathematics and physics they shared a passion for sport, particularly football. Harald, the better player, won a silver medal at the 1908 Olympics as a member of the Danish football team that lost to England in the final. Also regarded by many to be intellectually more gifted, he gained a doctorate in mathematics a year before Niels received his in physics in May 1911. Their father, however, always maintained that his eldest son was 'the special one in the family'.10
Dressed in white tie and tails as custom demanded, Bohr began the public defence of his doctoral thesis. It lasted just 90 minutes, the shortest on record. One of the two examiners was his father's friend Christian Christiansen. He regretted that no Danish physicist 'was well enough informed about the theory of metals to be able to judge a dissertation on the subject'.11 Nevertheless, Bohr was awarded his doctorate and sent copies of the thesis to men like Max Planck and Hendrik Lorentz. When no one replied he knew it had been a mistake to send it without first having it translated. Instead of German or French, which many leading physicists spoke fluently, Bohr decided on an English translation and managed to convince a friend to produce one.
Whereas his father had chosen Leipzig and his brother Göttingen, German universities being the traditional place for high-flying Danes to complete their education, Bohr chose Cambridge University. The intellectual home of Newton and Maxwell was for him 'the centre of physics'.12 The translated thesis would be his calling card. He hoped that it would lead to a dialogue with Sir Joseph John Thomson, the man he described later as 'the genius who showed the way for everybody'.13
![]()
After a lazy summer of sailing and hiking, Bohr arrived in England at the end of September 1911 on a one-year scholarship funded by Denmark's famous Carlsberg brewery. 'I found myself rejoicing this morning, when I stood outside a shop and by chance happened to read the address "Cambridge" over the door', he wrote to his fiancée Margrethe Nørland.14 The letters of introduction and the Bohr name led to a warm welcome from the university's physiologists who remembered his late father. They helped him find a small two-room flat on the edge of town and he was kept 'very busy with arrangements, visits and dinner parties'.15 But for Bohr it was his meeting with Thomson, J.J. to his friends and students alike, which soon preyed on his mind.
A bookseller's son from Manchester, Thomson had been elected the third head of the Cavendish Laboratory in 1884 within a week of his 28th birthday. He was an unlikely choice, after James Clerk Maxwell and Lord Rayleigh, to lead the prestigious experimental research facility, and not just because of his youth. 'J.J. was very awkward with his fingers,' one of his assistants later admitted, 'and I found it necessary not to encourage him to handle the instruments.'16 Yet if the man who won the Nobel Prize for discovering the electron lacked a delicate touch, others testified to Thomson's 'intuitive ability to comprehend the inner working of intricate apparatus without the trouble of handling it'.17
The polite manner of the slightly dishevelled Thomson, the epitome of the absent-minded professor in his round-rimmed glasses, tweed jacket and winged collar, helped calm Bohr's nerves when they first met. Eager to impress, he had walked into the professor's office clutching his thesis and a book written by Thomson. Opening the book, Bohr pointed to an equation and said, 'This is wrong.'18 Though not used to having his past mistakes paraded before him in such a forthright manner, J.J. promised to read Bohr's thesis. Placing it on top of a stack of papers on his overcrowded desk, he invited the young Dane to dinner the following Sunday.
Initially delighted, as the weeks passed and the thesis remained unread, Bohr became increasingly anxious. 'Thomson,' he wrote to Harald, 'has so far not been easy to deal with as I thought the first day.'19 Yet his admiration for the 55-year-old was undiminished: 'He is an excellent man, incredibly clever and full of imagination (you should hear one of his elementary lectures) and extremely friendly; but he is so immensely busy with so many things, and he is so absorbed in his work that it is very difficult to get to talk to him.'20 Bohr knew that his poor English did not help. So with the aid of a dictionary he began reading The Pickwick Papers as he fought to overcome the language barrier.
Early in November, Bohr went to see a former student of his father's who was now the professor of physiology at Manchester University. During the visit, Lorrain Smith introduced him to Ernest Rutherford, who had just returned from a physics conference in Brussels.21 The charismatic New Zealander, he recalled years later, 'spoke with characteristic enthusiasm about the many new prospects in physical science'.22 After being regaled with a 'vivid account of the discussions at the Solvay meeting', Bohr left Manchester charmed and impressed by Rutherford - both the man and the physicist.23
![]()
On his first day, in May 1907, the new head of physics at Manchester University caused a stir as he searched for his new office. 'Rutherford went up three stairs at a time, which was horrible to us, to see a Professor going up the stairs like that', remembered a laboratory assistant.24 But within a few weeks the boundless energy and earthy no-nonsense approach of the 36-year-old had captivated his new colleagues. Rutherford was on his way to creating an exceptional research team whose success over the next decade or so would be unmatched. It was a group shaped as much by Rutherford's personality as his inspired scientific judgement and ingenuity. He was not only its head, but also its heart.
Born on 30 August 1871 in a small, single-storey wooden house in Spring Grove on New Zealand's South Island, Rutherford was the fourth of twelve children. His mother was a schoolteacher and his father ended up working in a flax mill. Given the harshness of life in the scattered rural community, James and Martha Rutherford did what they could to ensure that their children had a chance to go as far as talent and luck would carry them. For Ernest it meant a series of scholarships that took him to the other side of the world and Cambridge University.
When he arrived at the Cavendish to study under Thomson in October 1895, Rutherford was far from the exuberant and self-confident man he would become within a few years. The transformation began as he continued work started in New Zealand on the detection of 'wireless' waves, later called radio waves. In only a matter of months Rutherford developed a much-improved detector and toyed with the idea of making money from it. Just in time, he realised that exploiting research for financial gain in a scientific culture where patents were rare would harm the chances of a young man yet to make his reputation. As the Italian Guglielmo Marconi amassed a fortune that could have been his, Rutherford never regretted abandoning his detector to explore a discovery that had been front-page news around the world.
On 8 November 1895, Wilhelm Röntgen found that every time he passed a high-voltage electric current through an evacuated glass tube, some unknown radiation was causing a small paper screen coated with barium platinocyanide to glow. When Röntgen, the 50-year-old professor of physics at the University of Würzburg, was later asked what he had thought on discovering his mysterious new rays, he replied: 'I did not think; I investigated.'25For nearly six weeks, he did 'the same experiment over and over again to make absolutely certain that the rays actually existed'.26 He confirmed that the tube was the source of the strange emanation causing the fluorescence.27
Röntgen asked his wife Bertha to place her hand on a photographic plate while he exposed it to 'X-rays', as he called the unknown radiation. After fifteen minutes Röntgen developed the plate. Bertha was frightened when she saw the outlines of her bones, her two rings and the dark shadows of her flesh. On 1 January 1896, Röntgen mailed copies of his paper, 'A New Kind of Rays', together with photographs of weights in a box and the bones in Bertha's hand, to leading physicists in Germany and abroad. Within days, news of Röntgen's discovery and his amazing photographs spread like wildfire. The world's press latched on to the ghostly photograph revealing the bones in his wife's hand. Within a year, 49 books and over a thousand scientific and semi-popular articles on X-rays would be published.28
Thomson had begun studying the sinister-sounding X-rays even before an English translation of Röntgen's paper appeared in the weekly science journal Nature on 23 January. Engaged in investigating the conduction of electricity through gases, Thomson turned his attention to X-rays when he read that they turned a gas into a conductor. Quickly confirming the claim, he asked Rutherford to help measure the effects of passing X-rays through a gas. For Rutherford the work led to four published papers in the next two years that brought him international recognition. Thomson provided a brief note to the first, suggesting, correctly as it later proved, that X-rays, like light, were a form of electromagnetic radiation.
While Rutherford was busy conducting his experiments, in Paris the Frenchman Henri Becquerel was trying to discover whether phosphorescent substances, which glow in the dark, could also emit X-rays. Instead he found that uranium compounds emitted radiation whether they were phosphorescent or not. Becquerel's announcement of his 'uranic rays' aroused little scientific curiosity and no newspapers clamoured to report his discovery. Only a handful of physicists were interested in Becquerel's rays for, like their discoverer, most believed that only uranium compounds emitted them. However, Rutherford decided to investigate the effects of 'uranic rays' on the electrical conductivity of gases. It was a decision he later described as the most important of his life.
Testing the penetration of uranium radiation using wafer-thin layers of 'Dutch metal', a copper-zinc alloy, Rutherford found that the amount of radiation detected depended on the number of layers used. At a certain point, adding further layers had little effect in reducing the intensity of radiation, but then surprisingly it began to fall once again as more layers were added. After repeating the experiment with different materials and finding the same general pattern, Rutherford could offer only one explanation. Two types of radiation were being emitted, and he called them alpha and beta rays.
When the German physicist Gerhard Schmidt announced that thorium and its compounds also emitted radiation, Rutherford compared it with alpha and beta rays. He found the thorium radiation to be more powerful and concluded that 'rays of a more penetrative kind were present'.29 These were later called gamma rays.30 It was Marie Curie who introduced the term 'radioactivity' to describe the emission of radiation and who labelled substances that emitted 'Becquerel rays' as 'radioactive'. She believed that since radioactivity was not confined to uranium alone, it must be an atomic phenomenon. It set her on the path to discovering, with her husband Pierre, the radioactive elements radium and polonium.
In April 1898, as Curie's first paper was published in Paris, Rutherford learned that there was a vacant professorship at McGill University in Montreal, Canada. Although acknowledged as a pioneer in the new field of radioactivity, Rutherford put his name forward with little expectation of being appointed, despite a glowing letter of recommendation from Thomson. 'I have never had a student with more enthusiasm or ability for original research than Mr Rutherford,' wrote Thomson, 'and I am sure if elected, he would establish a distinguished school of physics at Montreal.'31 He concluded: 'I should consider any institution fortunate that secured the services of Mr Rutherford as professor of physics.' After a stormy voyage, Rutherford, just turned 27, arrived in Montreal at the end of September and stayed for the next nine years.
Even before he left England he knew that he was 'expected to do a lot of original work and to form a research school to knock the shine out of the Yankees!'32 He did just that, beginning with the discovery that the radioactivity of thorium decreased by half in one minute and then by half again in the next. After three minutes it had fallen to an eighth of its original value.33 Rutherford called this exponential reduction of radioactivity the 'half-life', the time taken for the intensity of radiation emitted to fall by half. Each radioactive element had its own characteristic half-life. Then came the discovery that would earn him the professorship in Manchester and a Nobel Prize.
In October 1901, Rutherford and Frederick Soddy, a 25-year-old British chemist at Montreal, began a joint study of thorium and its radiation and were soon faced with the possibility that it could be turning into another element. Soddy recalled how he stood stunned at the thought and let slip, 'this is transmutation'. 'For Mike's sake, Soddy, don't call it transmutation', warned Rutherford. 'They'll have our heads off as alchemists.'34
The pair were soon convinced that radioactivity was the transformation of one element into another through the emission of radiation. Their heretical theory was met with widespread scepticism but the experimental evidence quickly proved decisive. Their critics had to discard long-cherishe beliefs in the immutability of matter. No longer an alchemist's dream, but a scientific fact: all radioactive elements did spontaneously transform into other elements, the half-life measuring the time it took for half the atoms to do so.
'Youthful, energetic, boisterous, he suggested anything but the scientist', is how Chaim Weizmann, later the first president of Israel but then a chemist at Manchester University, remembered Rutherford. 'He talked readily and vigorously on any subject under the sun, often without knowing anything about it. Going down to the refectory for lunch, I would hear the loud, friendly voice rolling up the corridor.'35 Weizmann found Rutherford 'devoid of any political knowledge or feelings, being entirely taken up with his epoch-making scientific work'.36 At the centre of that work lay his use of the alpha particle to probe the atom.
But what exactly was an alpha particle? It was a question that had long vexed Rutherford after he discovered that alpha rays were in fact particles with a positive charge that were deflected by strong magnetic fields. He believed that an alpha particle was a helium ion, a helium atom that had lost two electrons, but never said so publicly because the evidence was purely circumstantial. Now, almost ten years after discovering alpha rays, Rutherford hoped to find definitive proof of their true character. Beta rays had already been identified as fast-moving electrons. With the help of another young assistant, this time 25-year-old German Hans Geiger, Rutherford confirmed in the summer of 1908 what he had long suspected: an alpha particle was indeed a helium atom that had lost two electrons.
'The scattering is the devil', Rutherford had complained as he and Geiger tried to unmask the alpha particle.37 He had first noticed the effect two years earlier in Montreal when some alpha particles that had passed through a sheet of mica were slightly deflected from their straight-line trajectory, causing fuzziness on a photographic plate. Rutherford made a mental note to follow it up. Soon after arriving in Manchester, he had drawn up a list of potential research topics. Rutherford now asked Geiger to investigate one of those items - the scattering of alpha particles.
Together they devised a simple experiment that involved counting scintillations, tiny flashes of light produced by alpha particles when they strike a paper screen coated with zinc sulphide, after passing through a thin sheet of gold foil. Counting scintillations was an arduous task, with long hours spent in total darkness. Luckily, according to Rutherford, Geiger was 'a demon at the work and could count at intervals for a whole night without disturbing his equanimity'.38 He found that alpha particles either passed straight through the gold foil or were deflected by one or two degrees. This was as expected. However, surprisingly, Geiger also reported finding a few alpha particles 'deflected through quite an appreciable angle'.39
Before he could fully consider the implications, if any, of Geiger's results, Rutherford was awarded the Nobel Prize for chemistry for discovering that radioactivity was the transformation of one element into another. For a man who regarded 'all science as either physics or stamp collecting', he appreciated the funny side of his own instant transmutation from physicist to chemist.40 After returning from Stockholm with his prize, Rutherford learnt to evaluate the probabilities associated with different degrees of alpha particle scattering. His calculations revealed that there was a very small chance, almost zero, that an alpha particle passing through gold foil would undergo multiple scatterings resulting in an overall large-angle deflection.
It was while Rutherford was preoccupied with these calculations that Geiger spoke to him about assigning a project to Ernest Marsden, a promising undergraduate. 'Why not,' said Rutherford, 'let him see if any alpha particles can be scattered through a large angle?'41 He was surprised when Marsden did. As the search continued at ever-larger angles, there should have been none of the tell-tale flashes of light that Marsden had seen, signalling alpha particles crashing into the zinc sulphide screen.
As Rutherford struggled to make sense of 'the nature of the huge electric or magnetic forces which could turn aside or scatter a beam of alpha particles', he asked Marsden to check if any were reflected backwards.42 Not expecting him to find anything, he was utterly astonished when Marsden discovered alpha particles bouncing off the gold foil. 'It was,' Rutherford said, 'almost as incredible as if you had fired a 15-inch shell at a piece of tissue paper and it came back and hit you.'43
Geiger and Marsden set about making comparative measurements using different metals. They found that gold scattered backwards almost twice as many alpha particles as silver and twenty times more than aluminium. Only one alpha particle in every 8,000 bounced off a sheet of platinum. When they published these and other results in June 1909, Geiger and Marsden simply recounted the experiments and stated the facts without further comment. A baffled Rutherford brooded for the next eighteen months as he tried to think his way through to an explanation.
The existence of atoms had been a matter of considerable scientific and philosophical debate throughout the nineteenth century, but by 1909 the reality of atoms had been established beyond any reasonable doubt. The critics of atomism were silenced by the sheer weight of evidence against them, two key pieces of which were Einstein's explanation of Brownian motion and its confirmation, and Rutherford's discovery of the radioactive transformation of elements. After decades of argument, in which many eminent physicists and chemists had denied its existence, the most favoured representation of the atom to emerge was the so-called 'plum pudding' model put forward by J.J. Thomson.
In 1903 Thomson suggested that the atom was a ball of massless, positive charge in which were embedded like plums in a pudding the negatively-charged electrons he had discovered six years earlier. The positive charge would neutralise the repulsive forces between the electrons that would otherwise tear the atom apart.44 For any given element, Thomson envisaged these atomic electrons to be uniquely arranged in a set of concentric rings. He argued that it was the different number and distribution of electrons in gold and lead atoms, for example, which distinguished the metals from one another. Since all the mass of a Thomson atom was due to the electrons it contained, it meant there were thousands in even the lightest atoms.
Exactly one hundred years earlier, in 1803, the English chemist John Dalton first put forward the idea that atoms of every element were uniquely characterised by their weight. With no direct way of measuring atomic weights, Dalton determined their relative weights by examining the proportions in which different elements combined to form various compounds. First he needed a benchmark. Hydrogen being the lightest known element, Dalton assigned it an atomic weight of one. The atomic weights of all the other elements were then fixed relative to that of hydrogen.
Thomson knew his model was wrong after studying the results of experiments involving the scattering of X-rays and beta particles by atoms. He had overestimated the number of electrons. According to his new calculations, an atom could not have more electrons than prescribed by its atomic weight. The precise number of electrons in the atoms of the different elements was unknown, but this upper limit was quickly accepted as a first step in the right direction. The hydrogen atom with an atomic weight of one could have only one electron. However, the helium atom with an atomic weight of four could have two, three, or even four electrons, and so on for the other elements.
This drastic reduction in electron numbers revealed that most of the weight of an atom was due to the diffuse sphere of positive charge. Suddenly, what Thomson had originally invoked as nothing more than a necessary artifice to produce a stable, neutral atom took on a reality of its own. But even this new, improved model could not explain alpha particle scattering and failed to pin down the exact number of electrons in a particular atom.
Rutherford believed that alpha particles were scattered by an enormously strong electric field within the atom. But inside J.J.'s atom, with its positive charge evenly distributed throughout, there was no such intense electric field. Thomson's atom simply could not send alpha particles hurtling backwards. In December 1910, Rutherford finally managed to 'devise an atom much superior to J.J.'s'.45 'Now,' he told Geiger, 'I know what the atom looks like!'46 It was nothing like Thomson's.
Rutherford's atom consisted of a tiny positively-charged central core, the nucleus, which contained virtually all the atom's mass. It was 100,000 times smaller than the atom, occupying only a minute volume, 'like a fly in a cathedral'.47 Rutherford knew that electrons inside an atom could not be responsible for the large deflection of alpha particles, so to determine their exact configuration around the nucleus was unnecessary. His atom was no longer the 'nice hard fellow, red or grey in colour, according to taste' that he once, tongue-in-cheek, said he had been brought up to believe in.48
Most alpha particles would pass straight through Rutherford's atom in any 'collision', since they were too far from the tiny nucleus at its heart to suffer any deflection. Others would veer off course slightly as they encountered the electric field generated by the nucleus, resulting in a small deflection. The closer they passed to the nucleus, the stronger the effect of its electric field and the greater the deflection from their original path. But if an alpha particle approached the nucleus head-on, the repulsive force between the two would cause it to recoil straight back like a ball bouncing off a brick wall. As Geiger and Marsden had found, such direct hits were extremely rare. It was, Rutherford said, 'like trying to shoot a gnat in the Albert Hall at night'.49
Rutherford's model allowed him to make definite predictions, using a simple formula he had derived, about the fraction of scattered alpha particles to be found at any angle of deflection. He did not want to present his atomic model until it had been tested by a careful investigation of the angular distribution of scattered alpha particles. Geiger undertook the task and found alpha particle distribution to be in total agreement with Rutherford's theoretical estimates.
On 7 March 1911, Rutherford announced his atomic model in a paper presented at a meeting of the Manchester Literary and Philosophical Society. Four days later, he received a letter from William Henry Bragg, the professor of physics at Leeds University, informing him that 'about 5 or 6 years ago' the Japanese physicist Hantaro Nagaoka had constructed an atom with 'a big positive centre'.50 Unknown to Bragg, Nagaoka had visited Rutherford the previous summer as part of a grand tour of Europe's leading physics laboratories. Less than two weeks after Bragg's letter, Rutherford received one from Tokyo. Nagaoka wrote offering his gratitude 'for the great kindness you showed me in Manchester' and pointing out that in 1904 he had proposed a 'Saturnian' model of the atom.51 It consisted of a large heavy centre surrounded by rotating rings of electrons.52
'You will notice that the structure assumed in my atom is somewhat similar to that suggested by you in your paper some years ago', acknowledged Rutherford in his reply. Though alike in some respects, there were significant differences between the two models. In Nagaoka's the central body was positively-charged, heavy and occupied most of the flat pancake-like atom. Whereas Rutherford's spherical model had an incredibly tiny positively-charged core that contained most of the mass, leaving the atom largely empty. However, both models were fatally flawed and few physicists gave them a second thought.
An atom with stationary electrons positioned around a positive nucleus would be unstable, because the electrons with their negative charge would be irresistibly pulled towards it. If they moved around the nucleus, like planets orbiting the sun, the atom would still collapse. Newton had shown long ago that any object moving in a circle undergoes acceleration. According to Maxwell's theory of electromagnetism, if it is a charged particle, like an electron, it will continuously lose energy in the form of electromagnetic radiation as it accelerates. An orbiting electron would spiral into the nucleus within a thousandth of a billionth of a second. The very existence of the material world was compelling evidence against Rutherford's nuclear atom.
He had long been aware of what appeared to be an intractable problem. 'This necessary loss of energy from an accelerated electron,' Rutherford wrote in his 1906 book Radioactive Transformations, 'has been one of the greatest difficulties met with in endeavouring to deduce the constitution of a stable atom.'53 But in 1911 he chose to ignore the difficulty: 'The question of the stability of the atom proposed need not be considered at this stage, for this will obviously depend upon the minute structure of the atom, and on the motion of the constituent charged part.'54
Geiger's initial testing of Rutherford's scattering formula had been quick and limited in scope. Marsden now joined him in spending most of the next year conducting a more thorough investigation. By July 1912 their results confirmed the scattering formula and the main conclusions of Rutherford's theory.55 'The complete check,' Marsden recalled years later, 'was a laborious but exciting task.'56 In the process they also discovered that the charge of the nucleus, taking into account experimental error, was about half the atomic weight. With the exception of hydrogen, with an atomic weight of one, the number of electrons in all other atoms had to be approximately equal to half the atomic weight. It was now possible to nail down the number of electrons in a helium atom, for example, as two, where previously it could have been as many as four. However, this reduction in the number of electrons implied that Rutherford's atom radiated energy even more strongly than had previously been suspected.
As Rutherford recounted tales from the first Solvay conference for Bohr's benefit, he failed to mention that in Brussels neither he nor anyone else discussed his nuclear atom.
![]()
Back in Cambridge, the intellectual rapport that Bohr sought with Thomson never happened. Years later, Bohr identified one possible reason for the failure: 'I had no great knowledge of English and therefore I did not know how to express myself. And I could say only that this is incorrect. And he was not interested in the accusation that it was not correct.'57 Infamous for neglecting papers and letters from students and colleagues alike, Thomson was also no longer actively engaged in electron physics.
Increasingly disenchanted, Bohr met Rutherford again at the Cavendish research students' annual dinner. Held in early December, it was a rowdy, informal affair with toasts, songs and limericks following a ten-course meal. Once again struck by the personality of the man, Bohr seriously began thinking about swapping Cambridge and Thomson for Manchester and Rutherford. Later that month he went to Manchester and discussed the possibility with Rutherford. A young man separated from his fiancée, Bohr desperately wanted something tangible to show for their year apart. Telling Thomson that he wanted 'to know something about radioactivity', Bohr was granted permission to leave at the end of the new term.58 'The whole thing was very interesting in Cambridge,' he admitted many years later, 'but it was absolutely useless.'59
With only four months left in England, Bohr arrived in Manchester in the middle of March 1912 to begin a seven-week course in the experimental techniques of radioactive research. With no time to lose, Bohr spent his evenings working on the application of electron physics to provide a better understanding of the physical properties of metals. With Geiger and Marsden among the instructors, he successfully completed the course and was given a small research project by Rutherford.
'Rutherford is a man whom one cannot be mistaken about,' Bohr wrote to Harald, 'he comes regularly to hear how things are going and talk about every little thing.'60 Unlike Thomson, who seemed to him unconcerned about the progress of his students, Rutherford was 'really interested in the work of all people who are around him'. He had an uncanny ability to recognise scientific promise. Eleven of his students, along with several close collaborators, would win the Nobel Prize. As soon as Bohr arrived in Manchester, Rutherford wrote to a friend: 'Bohr, a Dane, has pulled out of Cambridge and turned up here to get some experience in radioactive work.'61 Yet there was nothing in what Bohr had done to date to suggest that he was any different from the other eager young men in his laboratory, except the fact that he was a theorist.
Rutherford held a generally low opinion of theorists and never lost an opportunity to air it. 'They play games with their symbols,' he once told a colleague, 'but we turn out the real solid facts of Nature.'62 On another occasion when invited to deliver a lecture on the trends of modern physics, he replied: 'I can't give a paper on that. It would only take two minutes. All I could say would be that the theoretical physicists have got their tails up and it is time that we experimentalists pulled them down again!'63 Yet he had immediately liked the 26-year-old Dane. 'Bohr's different', he would say. 'He's a football player!'64
Late every afternoon, work in the laboratory stopped as the research students and staff gathered to chat over tea, cakes and slices of bread and butter. Rutherford would be there, sitting on a stool with plenty to say, whatever the subject. But most of the time the talk was simply of physics, particularly of the atom and radioactivity. Rutherford had succeeded in creating a culture where there was an almost tangible sense of discovery in the air, where ideas were openly exchanged and discussed in the spirit of co-operation, with no one afraid to speak - even a newcomer. At its centre was Rutherford, who Bohr knew was always prepared 'to listen to every young man, when he felt he had any idea, however modest, on his mind'.65 The only thing Rutherford could not stand was 'pompous talk'. Bohr loved to talk.
Unlike Einstein who spoke and wrote fluently, Bohr frequently paused as he struggled to find the right words to express himself, whether in Danish, English or German. When Bohr spoke, he was often only thinking aloud in search of clarity. It was during the tea breaks that he got to know the Hungarian Georg von Hevesy, who would win the 1943 Nobel Prize for chemistry for developing the technique of radioactive tracing that was to become a powerful diagnostic tool in medicine, with widespread applications in chemical and biological research.
Strangers in a strange country, speaking a language that both had yet to master, the pair formed an easy friendship that lasted a lifetime. 'He knew how to be helpful to a foreigner', Bohr said as he recalled how Hevesy, only a few months older, helped him ease into the life of the laboratory.66 It was during their conversations that Bohr first began to focus on the atom, as Hevesy explained that so many radioactive elements had been discovered that there was not enough room to accommodate them all in the periodic table. The very names given to these 'radioelements', spawned in the process of radioactive disintegration of one atom into another, captured the sense of uncertainty and confusion surrounding their true place within the atomic realm: uranium-X, actinium-B, thorium-C. But there was, Hevesy told Bohr, a possible solution proposed by Rutherford's former Montreal collaborator, Frederick Soddy.
In 1907 it was discovered that two elements produced during radioactive decay, thorium and radiothorium, were physically different but chemically identical. Every chemical test they were subjected to failed to tell them apart. During the next few years, other such sets of chemically inseparable elements were discovered. Soddy, now based at Glasgow University, suggested that the only difference between these new radioelements and those with which they shared 'complete chemical identity' was their atomic weight.67 They were like identical twins whose only distinguishing feature was a slight difference in weight.
Soddy proposed in 1910 that chemically inseparable radioelements, 'isotopes' as he later called them, were just different forms of the same element and should therefore share its slot in the periodic table.68 It was an idea at odds with the existing organisation of elements within the periodic table, which listed them in order of increasing atomic weight, with
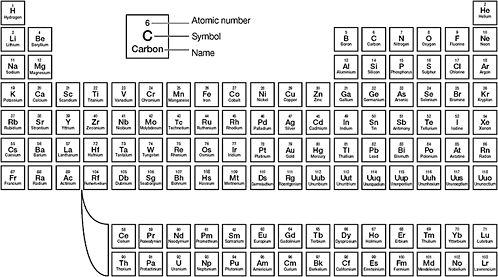
Figure 5: The periodic table
hydrogen first and uranium last. Yet the fact that radiothorium, radioactinium, ionium, and uranium-X were all chemically identical to thorium was strong evidence in favour of Soddy's isotopes.69
Until his chats with Hevesy, Bohr had shown no interest in Rutherford's atomic model. But he now had an idea: it was not enough to distinguish between the physical and chemical properties of an atom; one had to differentiate between nuclear and atomic phenomena. Ignoring the problem of its inevitable collapse, Bohr took Rutherford's nuclear atom seriously as he tried to reconcile isotopes with the use of atomic weights to order the periodic table. 'Everything,' he said later, 'then fell into line.'70
Bohr understood that it was the charge of the nucleus in Rutherford's atom that fixed the number of electrons it contained. Since an atom was neutral, possessing no overall charge, he knew that the positive charge of the nucleus had to be balanced by the combined negative charge of all its electrons. Therefore the Rutherford model of the hydrogen atom must consist of a nuclear charge of plus one and a single electron with a charge of minus one. Helium with a nuclear charge of plus two must have two electrons. This increase in nuclear charge coupled to a corresponding number of electrons led all the way up to the then heaviest-known element, uranium, with a nuclear charge of 92.
For Bohr the conclusion was unmistakable: it was nuclear charge and not atomic weight that determined the position of an element within the periodic table. From here he took the short step to the concept of isotopes. It was Bohr, not Soddy, who recognised nuclear charge as being the fundamental property that tied together different radioelements that were chemically identical but physically different. The periodic table could accommodate all the radioelements; they just had to be housed according to nuclear charge.
At a stroke, Bohr was able to explain why Hevesy had been unable to separate lead and radium-D. If the electrons determined the chemical properties of an element, then any two with the same number and arrangement of electrons would be identical twins, chemically inseparable. Lead and radium-D had the same nuclear charge, 82, and therefore the same number of electrons, 82, resulting in 'complete chemical identity'. Physically they were distinct because of their different nuclear masses: approximately 207 for lead and 210 for radium-D. Bohr had worked out that radium-D was an isotope of lead and as a result it was impossible to separate the two by any chemical means. Later, all isotopes were labelled with the name of the element of which they were an isotope and their atomic weight. Radium-D was lead-210.
Bohr had grasped the essential fact that radioactivity was a nuclear and not an atomic phenomenon. It allowed him to explain the process of radioactive disintegration in which one radioelement decayed into another with the emission of alpha, beta or gamma radiation as a nuclear event. Bohr realised that if radioactivity originated in the nucleus, then a uranium nucleus with a charge of plus 92 transmuting into uranium-X by emitting an alpha particle lost two units of positive charge, leaving behind a nucleus with a charge of plus 90. This new nucleus could not hold on to all of the original 92 atomic electrons, quickly losing two to form a new neutral atom. Every new atom formed as the product of radioactive decay immediately either acquires or loses electrons so as regain its neutrality. Uranium-X with a positive nuclear charge of 90 is an isotope of thorium. They both 'possessed the same nuclear charge and differed only in the mass and intrinsic structure of the nucleus', explained Bohr.71 It was the reason why those who tried, failed to separate thorium, with an atomic weight of 232, and 'uranium-X', thorium-234.
His theory of what was happening at the nuclear level in radioactive disintegration implied, Bohr said later, 'that by radioactive decay the element, quite independently of any change in its atomic weight, would shift its place in the periodic table by two steps down or one step up, corresponding to the decrease or increase in the nuclear charge accompanying the emission of alpha or beta rays, respectively'.72 Uranium decaying with the emission of an alpha particle into thorium-234 ended up two places further back in the periodic table.
Beta particles, being fast-moving electrons, have a negative charge of minus one. If a nucleus emits a beta particle, its positive charge increases by one - as if two particles, one positive and the other negative, that existed in harmony as a neutral pair had been ripped apart with the ejection of the electron, leaving behind its positive partner. The new atom produced by beta decay has a nuclear charge that is one greater than the disintegrating atom, moving it one place to the right in the periodic table.
When Bohr took his ideas to Rutherford he was warned about the danger of 'extrapolating from comparatively meagre experimental evidence'.73 Surprised by this muted reception, he attempted to convince Rutherford 'that it would be the final proof of his atom'.74 He failed. Part of the problem lay in Bohr's inability to express his ideas clearly. Rutherford, preoccupied with writing a book, did not make the time to fully grasp the significance of what Bohr had done. Rutherford believed that although alpha particles were emitted from the nucleus, beta particles were just atomic electrons somehow ejected from a radioactive atom. Despite Bohr's trying on five separate occasions to persuade him, Rutherford hesitated in following his logic all the way to its conclusion.75 Sensing that Rutherford was by now becoming 'a bit impatient' with him and his ideas, Bohr decided to let the matter rest.76 Others did not.
Frederick Soddy soon spotted the same 'displacement laws' as Bohr, but unlike the young Dane, he was able to publish his research without first having to seek approval of a superior. Nobody was surprised that Soddy was at the forefront of these breakthroughs. But no one could have guessed that an eccentric 42-year-old Dutch lawyer would introduce an idea of fundamental importance. In July 1911, in a short letter to the journal Nature, Antonius Johannes van den Broek speculated that the nuclear charge of a particular element is determined by its place in the periodic table, its atomic number, not its atomic weight. Inspired by Rutherford's atomic model, van den Broek's idea was based upon various assumptions that turned out to be wrong, such as nuclear charge being equal to half the atomic weight of the element. Rutherford was suitably annoyed that a lawyer should publish 'a lot of guesses for fun without sufficient foundation'.77
Having failed to gain any support, on 27 November 1913 in another letter to Nature, van den Broek dropped the assumption that the nuclear charge was equal to half the atomic weight. He did so after the publication of the extensive study by Geiger and Marsden into alpha particle scattering. A week later, Soddy wrote to Nature explaining that van den Broek's idea made clear the meaning of the displacement laws. Then came an endorsement from Rutherford: 'The original suggestion of van den Broek that the charge on the nucleus is equal to the atomic number and not to half the atomic weight seems to me very promising.' He was writing in praise of van den Broek's proposal a little more than eighteen months after advising Bohr against pursuing similar ideas.
Bohr never complained that he had missed out on being the first to publish the concept of atomic number, or those ideas that won Soddy the Nobel Prize for chemistry in 1921, due to Rutherford's lack of enthusiasm.78 'The confidence in his judgement,' Bohr fondly remembered, 'and our admiration for his powerful personality was the basis for the inspiration felt by all in his laboratory, and made us all try our best to deserve the kind and untiring interest he took in the work of everyone.'79 In fact, Bohr continued to regard an approving word from Rutherford as 'the greatest encouragement for which any of us could wish'.80 The reason why he could afford to be so generous, when others would have been left feeling disappointed and bitter, was what happened next.
![]()
After Rutherford dissuaded him from publishing his innovative ideas, by chance Bohr came across a recently published paper that grabbed his attention.81 It was the work of the only theoretical physicist on Rutherford's staff, Charles Galton Darwin, the grandson of the great naturalist. The paper concerned the energy lost by alpha particles as they passed through matter rather than being scattered by atomic nuclei. It was a problem that J.J. Thomson had originally investigated using his own atomic model, but which Darwin now re-examined on the basis of Rutherford's atom.
Rutherford had developed his atomic model using the large-angle alpha particle scattering data gathered by Geiger and Marsden. He knew that atomic electrons could not be responsible for such large-angle scattering and so ignored them. In formulating his scattering law that predicted the fraction of scattered alpha particles to be found at any angle of deflection, Rutherford had treated the atom as a naked nucleus. Afterwards he simply placed the nucleus at the centre of the atom and surrounded it with electrons without saying anything about their possible arrangement. In his paper, Darwin adopted a similar approach when he ignored any influence that the nucleus may have exerted on the passing alpha particles and concentrated solely on the atomic electrons. He pointed out that the energy lost by an alpha particle as it passed through matter was due almost entirely to collisions between it and atomic electrons.
Darwin was unsure how electrons were arranged inside Rutherford's atom. His best guess was that they were evenly distributed either throughout the atom's volume or over its surface. His results depended only on the size of the nuclear charge and the atom's radius. Darwin found that his values for various atomic radii were in disagreement with existing estimates. As he read this paper, Bohr quickly identified where Darwin had gone wrong. He had mistakenly treated the negatively-charged electrons as if they were free, instead of being bound to the positively-charged nucleus.
Bohr's greatest asset was his ability to identify and exploit failures in existing theory. It was a skill that served him well throughout his career, as he started much of his own work from spotting errors and inconsistencies in that of others. On this occasion, Darwin's mistake was Bohr's point of departure. While Rutherford and Darwin had considered the nucleus and the atomic electrons separately, each ignoring the other component of the atom, Bohr realised that a theory that succeeded in explaining how alpha particles interacted with atomic electrons might reveal the true structure of the atom.82 Any lingering disappointment over Rutherford's reaction to his earlier ideas was forgotten as he set about trying to rectify Darwin's mistake.
Bohr abandoned his usual practice of drafting letters even to his brother. 'I am not getting along badly at the moment,' Bohr reassured Harald, 'a couple of days ago I had a little idea with regard to understanding the absorption of alpha-rays (it happened in this way: a young mathematician here, C.G. Darwin (grandson of the real Darwin), has just published a theory about this problem, and I felt that it not only wasn't quite right mathematically (however, only slightly wrong) but very unsatisfactory in the basic conception, and I have worked out a little theory about it, which, even if it isn't much, perhaps may throw some light on certain things connected with the structure of atoms). I am planning to publish a little paper about it very soon.'83 Not having to go to the laboratory 'has been wonderfully convenient for working out my little theory', he admitted.84
Until he had put flesh onto the bare bones of his emerging ideas, the only person in Manchester whom Bohr was willing to confide in was Rutherford. Though surprised by the direction the Dane had taken, Rutherford listened and this time encouraged him to continue. With his approval, Bohr stopped going to the laboratory. He was under pressure, since his time in Manchester was almost up. 'I believe I have found out a few things; but it is certainly taking more time to work them out than I was foolish enough to believe at first', he wrote to Harald on 17 July, a month after first sharing his secret. 'I hope to have a little paper ready to show Rutherford before I leave, so I'm busy, so busy; but the unbelievable heat here in Manchester doesn't exactly help my diligence. How I look forward to talking to you!'85 He wanted to tell his brother that he hoped to fix Rutherford's flawed nuclear atom by turning it into the quantum atom.