The Human Side of Science: Edison and Tesla, Watson and Crick, and Other Personal Stories behind Science's Big Ideas (2016)


Hiroshima mushroom cloud. Courtesy of Isabella lynn Lee, from Wikimedia Commons.
WEAPONS OF MASS DESTRUCTION
What did you expect as a follow-up to Hubble? Not weapons. And it was certainly not what the scientists expected, either. But it brings up a critical issue.
Scientists who hypothesize and experiment to deepen our understanding of the universe aren't the only people on this planet—not by a long shot. When utilized by other people science's ideas exert an influence on the human condition. Sometimes the application of science is relatively minor—such as the use of non-polar Teflon molecules to keep polar grease molecules from sticking to pans. Sometimes it develops slowly, like the two hundred years from Bernoulli's principle to the Wright Brothers’ airplane. But in this case, the application of science to technology was both huge and quick. While it started from the extremely small, it grew to be enormously significant in only seven years, so hang on.
WHAT DID YOU DO IN THE WAR?
We know what Edwin Hubble did during World War I. Mostly, he trained in the United States, then spent a little time in France, where he earned his major's oak leaf. Did he think about the big picture of astronomy? Possibly.
For contrast, consider James Chadwick (1891-1974). Just before the war, Chadwick studied radioactivity under Ernest Rutherford at the University of Manchester in England, earned his MSc, then went to Berlin to study under Hans Geiger (of Geiger counter fame). When war broke out, Rutherford was sent to an internment camp in Ruhleben, where he remained for the duration of the war. Sounds pretty grim, eh? Not at all. With the cooperation of prison guards, Chadwick was allowed to set up a laboratory in a stable. He was able to obtain quantities of commercially available toothpaste that contained radioactive material, and to set up experiments with toothpaste and gum wrappers that contained aluminum foil.
When the war ended, Hubble worked on the universe's biggest challenges, using Mount Wilson's hundred-inch telescope; Chadwick returned to England to work on the smallest puzzles, namely, the nucleus, by following Rutherford to the Cavendish Laboratories at the University of Cambridge. Both Hubble and Chadwick were quite successful. Hubble's discovery of the expansion of the universe in 1929 revised the thinking of astronomers and opened whole new avenues of large-scale research. In 1932, Chadwick was able to demonstrate the existence of the subatomic particle called the neutron, the second of the components of atomic nuclei, and provided an extremely valuable new tool for probing the atom.
THE NEUTRON: SUSPECTED BUT VERY ELUSIVE
Rutherford had found evidence for the existence of the nucleus of the atom when he set up an experiment in which thin sheets of gold foil were bombarded by alpha particles, naturally occurring particles in radioactive decay of certain elements (see the introduction). In 1920, he showed that the simplest atom of all, hydrogen, had only one positively charged particle in its nucleus. He named this particle the proton. On the other hand, heavier nuclei in the periodic table had masses that weren't simply multiples of their number of protons. Further, radioactive decay of some elements gave off particles other than the alpha. One of these turned out to be the electron.
What was a tiny electron doing in the nucleus with all its massive protons? Was there some massive particle in the atom's nucleus in addition to the proton, but with no charge to upset the atom's equal numbers of electrons and protons? We need some background here before we can understand Chadwick's work.
RUSH TO DISCOVERY
Almost as if it was a deadline-beating phenomenon, the years before the turn of the twentieth century featured fascinating last-minute discoveries:
1895: X-rays, discovered by Wilhelm Röntgen
1896: radioactivity, discovered by Antoine Becquerel
1897: the electron, discovered by J. J. Thomson
While each of these discoveries was interesting in its own right, let's just look at radioactivity to understand how it all shakes out:
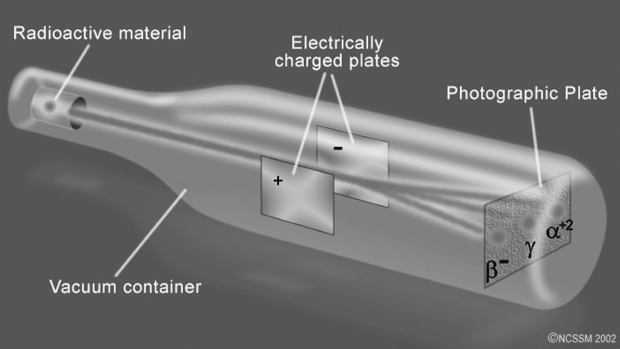
Radioactive decay products. Created by North Carolina School of Science and Mathematics. Licensed CC BY-NC-SA 4.0, used with permission.
· alpha (α)-particles are actually two protons and two neutrons stuffed together (charge = +2) and can be stopped by a shield as flimsy as a piece of paper; that's why Rutherford's experiments used extremely thin gold foil, since it could be made wafer-thin.
· beta (β)-particles are actually electrons (charge = -1) and cannot penetrate thin metal.
· gamma (γ)-particles are actually high-frequency light radiation (charge = 0) and can penetrate a thick lead shield.
While Hubble had used the world's biggest telescope to probe the large world of the nebulae by sensing visible light the nebulae emitted, experimenters investigating the nucleus had a much different problem. They had to shoot tiny invisible particles from radioactive decay and use cleverly designed detectors to find out what happened after these probes collided with the nucleus. Their results were confusing but interesting. Beta and gamma particles revealed a bit of information, and alpha particles were massive enough to penetrate into the heart of the atom, but their positive charge kept them from entering the nuclei because they were repelled by the positively charged nucleus. A new probe was needed.
BACK TO JAMES CHADWICK
Suspicion and reality came together in 1932. James Chadwick, in addition to serving as Rutherford's assistant director of research, managed to isolate and identify the particle that completed their understanding of the nucleus. This particle was slightly more massive than the proton and carried no electrical charge. Rutherford had already hypothesized the particle and named it the neutron. For his efforts, Chadwick was awarded the Nobel Prize in 1935. Just in case anyone might think the cartoon character Jimmy Neutron was named after Chadwick, it is true that Jimmy first appeared merely twenty-five years after Chadwick's death.
Besides completing the ideas about the atom's constituents, the neutron was almost immediately recognized as a valuable research tool. It had sufficient mass to penetrate an atom, and its electrical neutrality allowed it to penetrate the nucleus without being repelled. In short order, four different laboratories used neutrons to bombard heavier elements in order to produce elements with more neutrons in their nuclei. Technically these new versions of the original element are referred to as isotopes of that element because they have the same chemical properties as other isotopes of the same element, so they all occupy the same spot on the periodic table.
COMPETITION
Let's look at the competition between various laboratories as scientists endeavored to complete the understanding of the atomic nucleus.
The Cavendish Laboratories in Cambridge, England: The discoverer of the neutron was on staff here, and so the lab had a head start on the others.
The Radium Institute in Paris: Maria Skłodowska Curie's daughter Iréne Joliot-Curie and her husband, Frederic Joliot, produced radioactive isotopes by bombarding boron, magnesium, and aluminum with α-particles. For their work, they were awarded the Nobel Prize in Chemistry in 1935. When the couple tried neutron bombardment of uranium, the results were curious and confusing.
The University of Rome: Enrico Fermi's group had the most efficient neutron source and set out on the most ambitious program. Their goal was to bombard every element with neutrons and check for resulting radioactive isotopes. One of Fermi's innovations was to slow the neutrons down to facilitate their capture by nuclei. All went smoothly until they reached the largest naturally occurring element, uranium, atomic number 92. Colleagues thought he might have created a new element, one with atomic number 93, but Fermi was not so sure. In Fermi's words, “We did not know enough chemistry to separate the products from one another.”1
In 1934, German chemist Ida Tacke Noddack criticized Fermi's results because Fermi's group hadn't tested their products for elements lighter than lead. “It is conceivable that the nucleus breaks up into several large fragments, which would of course be isotopes of known elements but would not be neighbors of the irradiated element.”2 Since these projects were not in her direct field, she had no way to pursue them, but later developments were exactly along the lines she suggested.
Fermi won the 1938 Nobel Prize for his efforts. As you may recall from the chapter 10 discussion of Maja Einstein Winteler's decision to leave Italy because of anti-Semitic laws, Italy was becoming an unsafe place for Fermi's Jewish wife, Laura. The Fermis convinced the Mussolini government to let the whole family go to Sweden to collect the Nobel Prize. Once they got the award, they didn't return to Italy. Instead, they traveled directly from Stockholm to New York, arriving January 2, 1939. As they landed, Enrico said to Laura, “We have founded the American branch of the Fermi family.”3
The Kaiser Wilhelm Institute for Chemistry in Berlin-Dahlem: The team of Lise Meitner and Otto Hahn had been collaborating since 1907 and wound up with the most complete understanding of uranium bombardment of any of the groups. The connections get complex, so let's have a look at each member of the team before we see their enormously significant results.
OTTO HAHN (1879-1968)
Otto Hahn was the youngest of four children. As a youth, Hahn loved chemistry, he even performed experiments in the family laundry room. Although his father wanted him to study architecture, his desires for chemistry won out, and he wound up with a PhD in chemistry in 1901 from the University of Marburg. After postdoctoral studies at University College in London and at McGill University in Montreal with Ernest Rutherford, Hahn had found a specialty: radiochemistry. He managed to isolate several radioactive isotopes, and he received this comment from Rutherford: “Hahn has a special nose for discovering new elements.”4
Later in 1906, Hahn returned to Germany to work with Emil Fischer at the University of Berlin, and, within a few months, he discovered two new radioactive isotopes using extremely unsophisticated equipment. A year later, Hahn met the physicist Lise Meitner. They began a lifelong friendship and an extremely fruitful scientific collaboration.
THE PARTNERSHIP'S OTHER HALF: LISE MEITNER (1878-1968)
As the third of eight children of wealthy parents, Lise (originally Elise) Meitner was tutored to bring her education level up to that of her brothers. She excelled in physics and was admitted to the University of Vienna, where she was inspired by Ludwig Boltzmann. Her nephew, Otto Robert Frisch, wrote that “Boltzmann gave her the vision of physics as a battle for ultimate truth, a vision she never lost.”5
In 1905, Meitner became the second woman to earn a PhD from the University of Vienna. Her thesis discussed both theoretical analysis and experimental determinations, which were a hallmark of her entire career. Thanks to family financial backing, Meitner went to Berlin and was allowed to attend Max Planck's lectures, a most uncommon occurrence for a woman. A year later, she became Planck's (unpaid) assistant and sought a place to perform laboratory experiments. Meitner approached Professor Heinrich Rubens, who offered her space in his physics lab but mentioned that chemistry professor Otto Hahn had indicated an interest in collaborating with her. The shy and reserved Meitner thus had a choice between working with a substantially older physicist or a chemist of her own age. She had worked with radioactivity at Vienna, so she had a headstart in that area. She chose to work with Hahn.
An early difficulty was that women weren't allowed in the chemistry labs, allegedly because of safety—“their hair might catch fire.” One wonders about some of the men with their full beards. The solution was to use a basement room, which had been a former carpenter's workshop with an outside entrance. A restaurant down the street provided the nearest toilet, but Meitner was determined and stuck it out. With Hahn performing the tedious chemical separations and Meitner making the physical measurements and analyzing the physics, the team discovered several new radioactive isotopes and published two papers on β-particles.

Lise Meitner and Otto Hahn. From Wikimedia Commons, user Pieter Kuiper.
Within the next few years, many changes occurred, including the onset of World War I, the founding of a new institute, and the awarding of a full professorship for Meitner at the University of Berlin. In addition, Meitner connected with many other physicists of the day. For example, she taught a seminar with Leo Szilárd in 1930 and corresponded with Albert Einstein about physics problems. Einstein was impressed. He referred to Meitner as “Our Marie Curie.”6
FINALLY, NEUTRON BOMBARDMENT
Although Meitner and Hahn's neutron source wasn't as efficient as Fermi's, they had Hahn's excellent skill with the chemistry as a bonus. Once an element is bombarded with a neutron, the product must be analyzed to ascertain which isotopes are actually present. Soon, the team was augmented by the addition of Fritz Strassmann, who had resigned from the Society of German Chemists when it was taken over by Nazis in 1933. Although Strassmann was blacklisted, Hahn and Meitner found a half-pay position for him at the University of Berlin in 1934, and he assisted greatly in their neutron bombardment project. (Strassmann and his wife also concealed a Jewish friend in their apartment for months during the war, at great personal risk.)
The Meitner/Hahn/Strassmann group identified sixteen radioactive isotopes with various half-lives, but they encountered a personal half-life problem close to home. Lise Meitner had been born Jewish but had converted to Christianity in 1908, as had two of her sisters. Further, as an Austrian citizen, she paid little attention to the accession of Hitler as Germany's chancellor. After 1933, many of Meitner's physics colleagues had already fled Germany, including Einstein, Szilárd and even her nephew Otto Frisch. But Meitner ignored political developments and focused on her work.
In fall 1938, several developments occurred in quick succession. The team was reaching the end of their bombardment project by working on uranium, and the Nazis annexed Austria (Anschluss). Meitner's friends urged her to escape and arranged to smuggle her to the Netherlands. Otto Hahn even gave her a diamond ring he had inherited from his mother in case she needed to bribe a border guard. She narrowly escaped and eventually wound up in Manne Siegbahn's laboratory in Stockholm, Sweden. (The ring's eventual whereabouts are unknown.) In November, Hahn traveled to Copenhagen. He and Meitner planned another set of experiments involving uranium in their neutron bombardment series. Hahn and Strassmann then carried out the experiments, they but were perplexed by the apparent presence of the much lighter element barium in the product.
In December, Hahn wrote to Meitner, “Perhaps you can come up with some sort of fantastic explanation, we know ourselves that it can't actually burst apart into barium.”7 Meitner advised them to continue with further tests, but their prior results were confirmed. Meitner's physicist nephew, Otto Robert Frisch, who worked with Niels Bohr in Copenhagen, was visiting his aunt over the Christmas holiday, and Meitner showed him the Hahn letter as they hiked together in the snow. With Meitner walking and Frisch on skis, they discussed the result and drew diagrams on the little pieces of paper that somehow seem to collect in scientists’ pockets.
Using Bohr's model of the nucleus as a liquid drop, they tried to visualize a neutron causing the wobbly drop to split. Frisch referred to this as fission, since the products were less massive than the original nucleus. Using Einstein's famous E = mc2, they realized that a substantial amount of energy would be released in the process. Meitner wrote back to Hahn, and they agreed to publish separate papers, with Hahn and Strassmann discussing the chemistry, and Meitner and Frisch the physics.
Here's an excerpt from Meitner and Frisch's paper: “O. Hahn and F. Strassmann have discovered a new type of nuclear reaction, the splitting into two smaller nuclei of the nuclei of uranium and thorium under neutron bombardment…. It can be shown by simple considerations that this type of nuclear reaction may be described in an essentially classical way like the fission of a liquid drop, and that the fission products must fly apart with kinetic energies of the order of hundred million electron-volts each.”8
THE WORD SPREADS
Otto Frisch wasted no time in explaining this result to Niels Bohr in Copenhagen, who understood it almost immediately. Not only was a substantial amount of energy released, but more neutrons might be generated, which might lead to more fission events, and more fission events…a chain reaction. Bohr arrived in the United States in January of 1939 for a conference at George Washington University on low-temperature physics and superconductivity. Addressing this group, Bohr ignored the stated conference topic and excitedly announced the fission experiment results. Comments were remarkably subdued. One physicist recalls a colleague whispering in his ear, “Perhaps we should not discuss this.”9
WAY MORE THAN DISCUSSION
A corner had been turned. It was clear that fission produced energy, but the questions of how the energy might be used in a practical sense and when it might be available were unknown. Even more importantly, who might control the energy and for what purpose were critical questions. The initial experiments were carried out in Germany, perhaps putting Germany in the driver's seat. Germany's military ambitions were on clear display, as its troops had just overrun the Sudetenland, and more German expansion was clearly in the wind. Bohr's native Denmark felt the pressure of the Nazi threat: many of those scientists at the conference had already fled Europe to avoid Hitler.
The next six years produced a whirlwind of scientific activity and even more technological and political commotion. To track these developments from a human perspective, we will focus on two individuals who played key roles: Enrico Fermi and Leo Szilárd.
ENRICO FERMI (1901-1954)
As we saw earlier, Fermi was in on the ground floor of neutron bombardment. He won the Nobel Prize in 1938 and promptly sailed for the United States. Fermi and his family arrived in early January 1939, just as the Meitner/Hahn papers were being published. When Bohr arrived in America for the conference, at which he revealed the fission news, he immediately sought out Fermi to tell him first. Fermi saw the possibilities but suggested a go-slow policy until their understanding was more complete.

Enrico Fermi (1901-1954). From Wikipedia, user Pieter Kuiper.
LEO SZILÁRD
A friend of both Einstein and Meitner from their Berlin days, Szilárd had been involved in many different aspects of physics. He conceived of the linear accelerator, cyclotron, and electron microscope and even filed a few patent applications, but he didn't follow through on any of these. In 1933, Szilárd saw the political handwriting on the wall and left Germany, settling in England. Learning of Rutherford's bombardment of lithium nuclei, Szilárd envisioned neutrons being given off in the process. These in turn could bombard other nuclei, leading to a chain reaction. He patented this idea and assigned it to the British Admiralty to ensure its secrecy.

Leo Szilárd (1898-1964). From Wikimedia Commons, user Panoptik~commonswiki.
Szilárd then worked on radioactive isotopes for medical treatment. Seeing a European war as inevitable, he immigrated to the United States in 1938 and conducted research at several universities across the country, winding up in New York in January 1939. When Szilárd heard about the uranium fission, it brought up the fear of Germany making a bomb using the chain reaction principle. Szilárd had no university affiliation, so he needed some strong academic connection. Fermi had a professorship at Columbia, so Szilárd decided to approach him.
THE ODD COUPLE
It's hard to imagine two people less similar. Fermi was a careful experimenter who always wanted to carry a project through to completion, whereas Szilárd nimbly jumped from one idea to another. Bernard T. Feld, Szilárd's research assistant, describes the difference between Fermi and Szilárd this way: “Fermi would not go from point A to point B unless he knew all he could about A and had reasonable assurances about B. Szilard would jump from point A to point D, then wonder why you were wasting your time with B and C.”10 Fermi was an early riser, sometimes getting up by five am to plan his day. Szilárd often slept in, then soaked in his bathtub for inspiration. Nevertheless, Szilárd was made a guest researcher at Columbia.
He and Fermi began the research that led to a practical nuclear fission reactor. They learned several things fairly quickly: the rare uranium isotope of atomic mass 235 undergoes fission when hit by slow neutrons, as opposed to the far more naturally occurring isotope 238, which cannot support a chain reaction; neutrons can be slowed by several substances, such as “heavy” water (which contains a large proportion of the more massive hydrogen ion, deuterium), or else highly purified carbon in the form of graphite. Both of these developments implied that large quantities of rare and costly materials would be necessary in order to make a fission reactor. Fermi continued his careful experimental work.
Szilárd however took a different route. He approached financiers and tried to raise money for private research. When that approach proved fruitless, he tried an entirely different tack. Starting in July, Szilárd paid several visits to his old friend Albert Einstein, who was vacationing in Peconic, Long Island. On the first visit, Szilárd and a college friend, physicist Eugene Wigner, got hopelessly lost. After going in circles for hours, they decided to ask a sunburnt boy of about seven if he knew where Einstein lived. “Of course I do,” he said and pointed the way. When they arrived, Einstein greeted them cordially, dressed in a white undershirt and rolled-up white trousers. When they outlined the fission developments, Einstein answered, “I haven't thought of that at all.”11 But he shared their concern, and the three of them drafted a letter that they planned to send to the Belgian ambassador and the US State Department. After a few more letter drafts and trips to Peconic, the following letter was generated:

From Wikimedia Commons, user Schutz.
The letter was delivered to President Franklin Delano Roosevelt by Alexander Sachs, a personal friend of the president's and a vice president of Wall Street's Lehman Corporation. Roosevelt said to him, “Alex, what you are after is to see the Nazis don't blow us up.” “Precisely,” said Sachs.12 The first step in a lengthy sequence was put in place: an advisory committee was set up.
THE MANHATTAN PROJECT
In June 1941, Roosevelt created the Office of Scientific Research and Development (OSRD) headed by Vannevar Bush (no relation to Presidents Bush 41 and Bush 43). In October, Roosevelt approved the fission effort, called the Manhattan Project, and agreed to coordinate with American efforts with those of the British. The British effort, code-named Tube Alloys, had a recent breakthrough investigating the amount of uranium 235 needed to sustain fission. British researchers Frisch and Peierls (more on Peierls later) found that the critical mass (amount required to have a self-sustaining chain reaction) of uranium required was approximately 1 kg (2.2 lbs). Does Frisch sound familiar? Otto Frisch was Lise Meitner's nephew, the one who had first named fission.
In December 1941, World War II began in earnest for the United States. Japan declared war on America and attacked Pearl Harbor; Germany declared war on the United States shortly thereafter. The Manhattan Project assumed even greater importance, since most everyone now agreed with Leo Szilárd. It was essential that the Americans beat the Germans to developing the fission bomb.
To maintain the security Szilárd had insisted on from the very beginning, work on the project was split up and carried out in different locations: Hanford, Washington; Oak Ridge, Tennessee; Long Island, New York; Chicago, Illinois; and Los Alamos, New Mexico. The idea was to keep the scientists isolated from each other to minimize communication so that no one could get a view of the entire project. Secrecy was so foreign to scientists’ usual procedure that it failed in many instances. Some instances were harmless, such as the efforts of Richard P. Feynman, who routinely opened the combination locks of absent colleagues’ safes to obtain needed reports. Feynman then left them a note telling them what he had done. Other security breaches were much more serious, such as the information supplied to the Russians by British physicist Klaus Fuchs.
Eventually, the Manhattan Project employed 130,000 people at thirty different sites in the United States, Canada, and the United Kingdom. Physicist J. Robert Oppenheimer was the director of the Los Alamos National Laboratory where the bombs were designed and built, and Brigadier General Leslie R. Groves from the Army Corps of Engineers was the overall project director. Fermi had a hand in every scientific aspect of the project. Szilárd was the project gadfly, making comments on every decision, sharing valuable insights, cajoling suppliers, and annoying almost everyone. His major clash was with General Groves. Both men had stocky builds, but the similarity ended there. Szilárd disliked authority and engineers, and Groves was both. Szilárd spoke with a Hungarian accent, had a difficult-to-trace background, and moved about erratically. Oddly, the security issue that so irritated Groves had been a hallmark of Szilárd's efforts. Nevertheless, Groves tried to have him removed from the project as a security risk, but failed when Szilárd turned over more details of his previous life and listed several references, including Einstein.
The first nuclear reactor was constructed in a former squash court under the stands at Stagg Field at the University of Chicago. Called CP-1 (for Chicago Pile-1), the reactor consisted of uranium pellets (six tons plus thirty-four tons of uranium oxide) and graphite blocks (four hundred tons), with control rods of cadmium, indium, and silver. When a uranium nucleus undergoes fission, the neutrons emitted are slowed by the graphite so that they can cause more fissions. Conversely, the control rods absorb neutrons so that they are unable to create more fissions.
On December 2, 1942, the construction of the CP-1 was complete, and all control rods were removed, save one. As the last control rod was withdrawn in increments, Fermi monitored the neutron emissions carefully until, finally, enough neutrons caused fissions to make the process self-sustaining—the pile had “gone critical.” After twenty-eight minutes of chain reaction, Fermi ordered the control rods replaced.
The test was a success. Director Arthur Compton sent a cable to James B. Conant, the chairman of the National Defense Research Committee, announcing the result in an ad hoc coded form:
Compton: The Italian navigator has landed in the New World.
Conant: How were the natives?
Compton: Very friendly.”13
Szilárd's view was a little different. He shook Fermi's hand and said that this would be regarded as a “black day in the history of mankind.”14 Now that a fission reactor had been demonstrated, the probability of building a bomb had become much more likely.
GERMAN NUCLEAR EFFORTS
Kurt Diebner headed the German nuclear fission research projects that began in April 1939. One of the major contributors was Werner Heisenberg, who had earned his PhD under Niels Bohr in Copenhagen. The research proceeded along similar lines as the Manhattan Project, but the Germans had a substantial headstart. By the middle of 1941, the Germans could see a path to building a fission reactor, but they realized it would require an enormous effort, both in personnel and resources.
In September 1941, a curious thing happened. Heisenberg accepted an invitation to speak at a German cultural institute in Copenhagen, in German-occupied Denmark. While he was there, Heisenberg met with Bohr, and they took a long walk together. Since there were no witnesses, many have speculated about the topic of their conversation and its possible relevance to nuclear fission research. Such speculations included a popular book (Brighter Than a Thousand Suns by Robert Jungk) and a Tony award-winning play, Copenhagen by Michael Frayn.
Whatever was said between the colleagues, Germany's leaders soon made a decision to concentrate the war effort on rockets and jet airplanes. The nuclear research was split up among nine institutes, the directors of which set their own objectives, so no coordinated effort was undertaken.
After the war ended, British intelligence rounded up many German scientists and interned them at Farm Hall, near Cambridge. Their quarters were bugged, and transcripts of the listening device recordings were scanned for information about how far along the German program had gotten. Here's a quote from Heisenberg: “The point is that the whole structure of the relationship between the scientist and the state in Germany was such that although we were not 100 percent anxious to do it, on the other hand we were so little trusted by the state that even if we had wanted to do it, it would not have been easy to get it through.”15
And here's a comment from Otto Hahn, chiding his colleagues who had worked on the project: “If the Americans have a uranium bomb then you're all second-raters.”16
AMERICAN FISSION BOMB
Indeed, the Americans did have a uranium bomb in 1945, but its development required more than two years of concentrated engineering and technological development beyond the reactor CP-1 stage. Besides the technical effort, a giant principle came into play here.
Let's look at a much milder example first. In the 1960s, one of us (AW) worked as an engineer for a company in the aerospace industry. The projects were long-range and research-oriented, but as the war in Vietnam heated up, the company investigated a new project outside our usual range. I was still busy on other matters, but a friend of mine was assigned to work on a proposal for a computer system for a new missile system. After learning the details of the project (he remembers a discussion of kill radius along the null range vector), my friend suggested that, rather than use a miniature computer to guide a proposed multiple warhead missile, the military should hire small people. He knew his sarcastic comment would get him either reassigned or fired, but he said it was a matter of ethics, which was more important than the job. My friend got a new assignment, and the project was dropped. No harm, no foul?
On the much larger scale of the devastating effects of the fission bomb, what were the ethical concerns of the scientists, engineers, technicians, military personnel, and their families? Could they, like my aerospace engineer friend, anticipate the harmful consequences of the Manhattan Project? Certainly, opinions and ethics varied all over the map. Further, the multiple sites and secrecy involved prevented any one person at any one time from having a clear picture at any one site of the whole project. But we do know about one person who had a bird's eye view, the ability to jump from A to D in a single bound, and the courage to speak up, to anybody: Leo Szilárd.
LEO SZILÁRD AGAIN
By mid-1943, the Nazis began suffering military reverses on every front. To forward thinkers like Leo Szilárd, the end of the war was in sight, even a year before D-Day. Szilárd was far more interested in nuclear power for peaceful purposes, and he worried about the destructive aspects of the bomb. When he later recalled the events of March 1944, he wrote: “Initially we were strongly motivated to produce the bomb because we feared the Germans would get ahead of us and the only way to prevent them from dropping bombs on us was to have bombs in readiness ourselves. But now, with the war won, it was not clear what we were working for.”17 As usual, Szilárd was thinking far in advance of anyone else, but other people began to catch up.
By early 1945, a rapid succession of events occurred that changed the world scene forever.
· March: Szilárd drafted a letter for Einstein to sign and send to President Roosevelt. The letter introduced Szilárd and requested a meeting between Roosevelt and Szilárd. The letter stated, “The terms of secrecy under which Dr. Szilard is working at present do not permit him to give me information about his work; however, I understand that he now is greatly concerned about the lack of adequate contact between scientists who are doing this work and members of your Cabinet who are responsible for formulating policy.”18

Used with permission from Sidney Harris.
· April: President Roosevelt died unexpectedly without seeing Szilárd. He was succeeded by Vice President Harry S. Truman, who had no knowledge of the Manhattan Project until after he was sworn in.
· May: Germany surrendered. This was known as VE Day, for victory in Europe. Some workers left the Manhattan Project, but the vast majority stayed to finish. Late in the month, Szilárd had an appointment at the White House. The president's appointments secretary, Matthew J. Connelly, said the president wanted former Senator James Byrnes of South Carolina to handle this matter. Szilárd agreed and traveled to Spartanburg in the former senator's home state, where he delivered his memorandum regarding the moral implications of the nuclear bomb. The meeting was unproductive. Byrnes's recollection was that Szilárd's “general demeanor and his desire to participate in policy-making made an unfavorable impression on me.” Szilárd said, “I was rarely as depressed as when we left Byrnes’ house.”19
· June: Arthur Compton, the director of the Met Lab, as the Chicago Manhattan Project site was called, wrote very insightfully about the political activities of his scientific colleagues: “The scientists will be held responsible, both by the public and their own consciences, for having faced the world with the existence of the new powers. The fact that the control has been taken out of their hands makes it necessary for them to plead the need for careful consideration and wise action to someone with authority to act. There is no other way in which they can meet their responsibility to society.”20
· July 17: Szilárd drafted A PETITION TO THE PRESIDENT OF THE UNITED STATES, which stated, in part:
In view of the foregoing, we, the undersigned, respectfully petition: first, that you exercise your power as Commander-in-Chief, to rule that the United States shall not resort to the use of atomic bombs in this war unless the terms which will be imposed upon Japan have been made public in detail and Japan knowing these terms has refused to surrender; second, that in such an event the question whether or not to use atomic bombs be decided by you in light of the considerations presented in this petition as well as all the other moral responsibilities which are involved.
Signed by Leo Szilárd and sixty-nine cosigners.
The petition was routed through official channels and had no discernable effect.
· July 16: Unknown to Szilárd, the first nuclear bomb was tested (code name: Trinity) at Los Alamos, New Mexico, at 5:45 am. Called “the Gadget,” the bomb detonated successfully. Military planners continued planning a strike on mainland Japan. President Truman's diary reflects his instructions: “I have told the Sec. of War, Mr. Stimson, to use it so that military objectives and soldiers and sailors are the targets and not women and children.”21
· August 6: The B-29 Enola Gay dropped a single nuclear fission bomb on the Japanese mainland city of Hiroshima at 8:15 am local time. The bomb detonated at an altitude of 19,000 feet and packed the destructive power of 16,000 tons of TNT. Achieving that effect with conventional bombs would require at least 2,000 fully loaded B-29s dropping 500-lb. bombs—simultaneously. The results were horrific: 70,000-80, 000 were killed instantly (20,000 soldiers) and another 70,000 injured. Three days later, a second bomb was dropped on Nagasaki, causing 22,000 to 75,000 deaths.
THE INHUMAN SIDE OF SCIENCE?
Even though the destruction experienced by the Japanese cities closely matched their predictions, many of the Manhattan Project scientists were horrified. The Bulletin of the Atomic Scientists was founded in 1945 by Manhattan Project scientists who “could not remain aloof to the consequences of their work.”22 The organization's early years chronicled the dawn of the nuclear age and the birth of the scientists’ movement, as told by the men and women who built the atomic bomb and then lobbied with both technical and humanist arguments for its abolition (see more at: http://thebulletin.org). On the cover of every issue is a Doomsday Clock, representing the scientists’ estimate of the countdown to possible global catastrophe.

Head-on collision of Physics, Technology, and Ethics. Used with permission from Sidney Harris.
IN THEIR OWN WORDS
Here are quotes from some of the people involved:
“…the greatest thing in history.”
—Harry S. Truman

Doomsday Clock. Courtesy of Sancheevi Sivakuma, from Wikimedia Commons.
“Japan was at the moment seeking some way to surrender with minimum loss of ‘face.’ It wasn't necessary to hit them with that awful thing.”
—General Dwight D. Eisenhower
“It is my opinion that the use of this barbarous weapon at Hiroshima and Nagasaki was of no material assistance in our war against Japan. The Japanese were already defeated and ready to surrender. My own feeling was that in being the first to use it, we had adopted an ethical standard common to the barbarians of the Dark Ages. I was taught not to make war in that fashion, and wars cannot be won by destroying women and children.”
—Admiral William D. Leahy, Former Chair of the Joint Chiefs of Staff
“I have no remorse about the making of the bomb and Trinity [the first test of an A-bomb]. That was done right. As for how we used it, I understand why it happened and appreciate with what nobility those men with whom I'd worked made their decision. But I do not have the feeling that it was done right. The ultimatum to Japan [the Potsdam Proclamation demanding Japan's surrender] was full of pious platitudes…. Our government should have acted with more foresight and clarity in telling the world and Japan what the bomb meant.”
—J. Robert Oppenheimer23
Since the nuclear bombs were dropped while many German scientists were still interned at Farm Hall, the transcripts reveal some of their reactions. Otto Hahn contemplated suicide, believing himself personally responsible for the many Japanese victims, while less than two weeks after the announcement Werner Heisenberg had figured out the process by which the bomb was built.
“So, you're the little lady who got us into all of this!” said President Harry S. Truman in 1946 when he met Lise Meitner.24
When asked to join the Manhattan Project in 1943, Lise Meitner replied, “I will have nothing to do with a bomb!”25
In 1947 Einstein told Newsweek magazine “Had I known that the Germans would not succeed in developing an atomic bomb, I would have done nothing.”26
Leo Szilárd sums it up: “I have been asked whether I would agree that the tragedy of the scientist is that he is able to bring about great advances in our knowledge, which mankind may then proceed to use for purposes of destruction. My answer is that this is not the tragedy of the scientist; it is the tragedy of mankind.”27
In 1947, Leo Szilárd switched from physics to biology and helped found the Salk Institute in La Jolla, California. Szilárd continued his eclectic brilliance in the new field and made many contributions to biological research. His insightful suggestions inspired one of his colleagues to remark that he loved being around Szilárd because “Leo got excited about something before it was true.”28 That colleague was James Watson, who figures prominently in the next chapter.