Ebola: How a People's Science Helped End an Epidemic - Paul Richards (2016)
Chapter 1. THE WORLD’S FIRST EBOLA EPIDEMIC
Ebola Virus Disease is a severe haemorrhagic fever caused by the crossover to humans of four species of filoviruses circulating in animal vectors in African forests and forest margins. Death rates range from 25 to 90 per cent.
In December 2013 the most deadly (Zaire) species of the virus caused human deaths in the forested part of south-eastern Guinea, and infection spread rapidly to two neighbouring countries, Liberia and Sierra Leone. In twenty or so previous African outbreaks Ebola had been localized and rapidly contained. In Upper West Africa the disease spread over a much larger area and travelled (in a few cases) between continents, thus becoming the world’s first Ebola epidemic.
The incubation period of the virus in humans ranges from two to twenty-one days. The course of the disease itself runs for about six days before organ collapse and death, or the onset of recovery.
The main symptoms of a first, dry phase (lasting about three days) include headache, fever and extreme tiredness, symptoms easily confused with those of malaria. In the second, wet phase the symptoms include vomiting, diarrhoea and bleeding from mouth, eyes and other orifices. No evidence has been found of airborne infection. Known infection pathways all involve direct contact with the body fluids of an Ebola patient. The virus enters through body orifices such as eyes and mouth, or through breaks in the skin.
Many dictionaries treat infection and contagion as more or less synonymous, but for Ebola a distinction can be drawn between the two terms. Ebola is highly infectious, in that the smallest droplet of blood, faeces or vomit is dangerous, as are sperm and breast milk after recovery, but the disease is not very contagious. The virus is fragile when exposed, and is not spread by casual contact. Infection occurs only in the wet stage, especially among those involved in nursing a victim or in disposing of a body.
Epidemiologists talk in terms of a reproduction number (Rn). This is the average number of subsequent infections per case. For an Ebola outbreak this number is not very high (typically about 2 - less than measles, for example). But those intimate with an Ebola patient are at high risk, since contact with body fluid is potentially lethal.
The crisis of the disease is the wet stage, a time of high risk for all attempting to care for the patient. Rehydration is important for successful patient care, but even a simple act such as refilling a drinking cup can infect the carer. Response agencies learnt to treat Ebola like cholera, also a disease with no antidote, where dehydration is a killer, and drips essential. But attaching a drip to a sick, fearful, restless Ebola patient is an extremely risky task for a nurse.
Cumbersome protective clothing is necessary (personal protective equipment, PPE). This inhibits movement, and limits the time a nurse or doctor can work in tropical heat. Learning how to put on and take off protective clothing safely is a crucial technique in Ebola epidemics. A tedious, inescapable routine must be followed to the letter, however pressing the demand. The protective dress terrified many patients, and fed rumours of germ warfare and body snatchers.
How bodies are handled in sickness and death is thus a central issue for treating Ebola patients and ending infection chains. Control calls for the rapid modification of techniques of the body on the part of all - whether lay or professional - who attempt to care for patients or handle dead bodies. In this lies the central theme of this book - how responders, and especially families and communities, acquired these modified techniques of the body.
Prior experience of Ebola
What was the state of knowledge about the disease when the Upper West African epidemic began?
Ebola first became known to science in the 1970s as the result of an outbreak in an isolated area adjacent to the Ebola river in the Democratic Republic of Congo (then known as Zaire). Early cases were associated with hunters, bushmeat and dead forest animals. Outbreaks were sometimes magnified by hospital infections.
An excellent general account of these earlier experiences is to be found in a book by two medical anthropologists, Barry and Bonny Hewlett.1 The Hewletts studied Ebola episodes in Uganda (2000) and Congo (2003) through first-hand ethnographic field research, and collated information on all other outbreaks up to 2007. Table 1.1 summarizes this information, supplemented by data on more recent episodes from World Health Organization (WHO) sources.
The single largest episode occurred in northern Uganda in 2000 (425 cases). Other outbreaks were mainly small, and confined to isolated forest or forest-edge communities. Infection pathways were often traced through hunters coming into contact with infected (sometimes dead) animals, such as chimpanzees and duikers. Fruit bats were identified as symptomless carriers of the virus.
The Uganda episode stands out not only as the largest outbreak in numbers of persons infected (prior to 2013), but also in being urban based. Previous episodes had been in remote, rural localities. The Gulu outbreak made clear some of the new challenges associated with outbreaks in more densely settled communities.
The response to the epidemic of 2013/15 had to face the challenge of how to respond to Ebola in both isolated rural conditions and also in densely crowded urban areas. There were large numbers of cases of Ebola in the capital cities of Guinea, Liberia and Sierra Leone. Each of these metropolitan regions contains populations in excess of one million. The spread of Ebola in urban slums such as the West Point district in Monrovia posed unprecedented challenges.
Table 1.1 Pre-2013 African outbreaks of Ebola virus disease (excluding single cases)
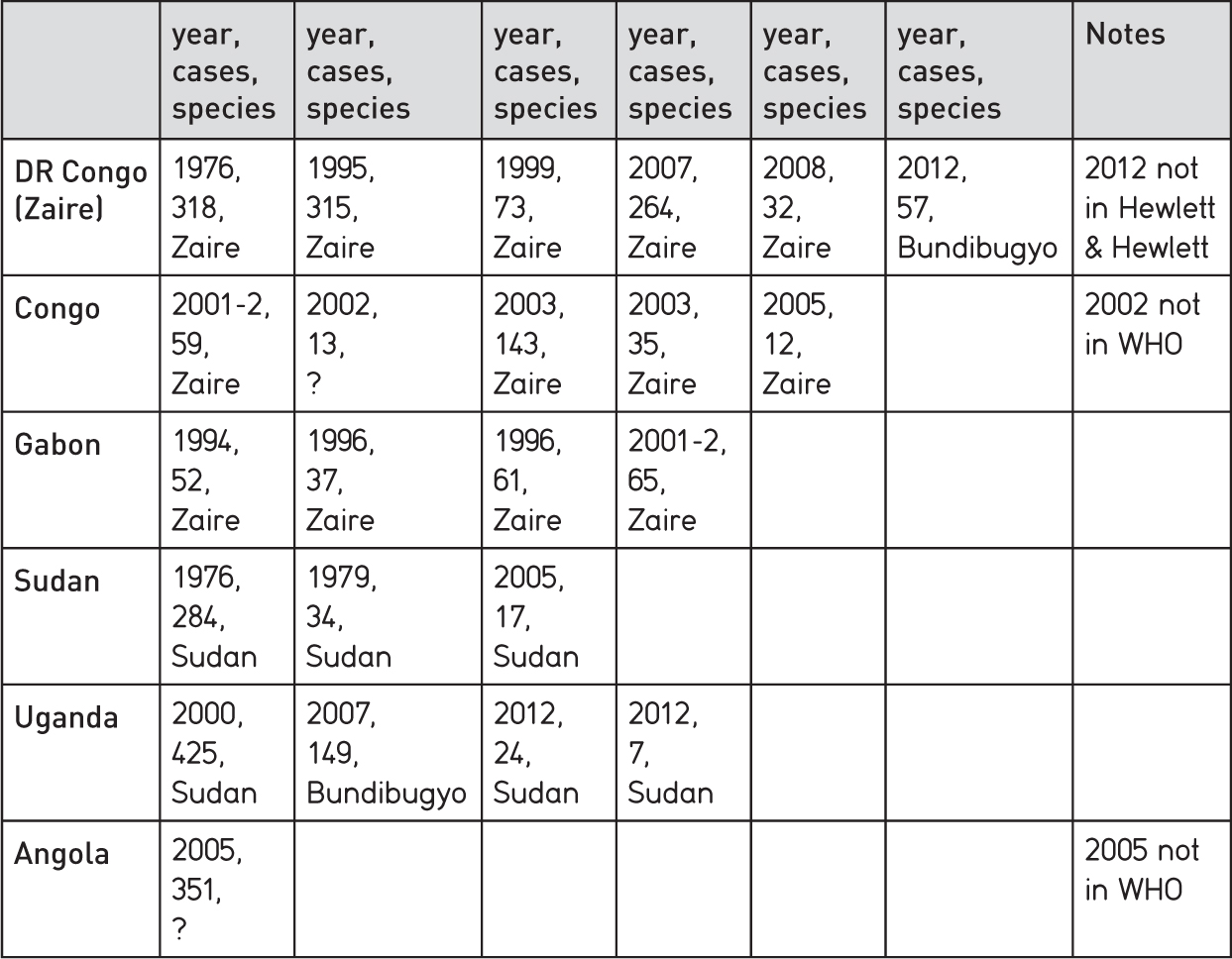
Sources: Hewlett and Hewlett (2008), Figure 1.2, p. 5; WHO: www.who.int/mediacentre/factsheets/Fs103_ebola; elsewhere Angola outbreak reported as Marburg virus disease, Andrew Meldrum, The Guardian, 5 April 2005.
The issue of how Ebola jumped the rural-urban divide is raised in the book by the Hewletts. They discuss a theory, widely believed locally, that the infection spread into Gulu town from Ugandan army personnel fighting Hutu rebels in DR Congo, perhaps through the repatriation of the body of a dead soldier. This seems unlikely, since the Gulu outbreak was caused by the Sudan not the Zaire species of the Ebola virus. Even so, we do now know from Upper West Africa that long-distance movements by members of highly mobile occupational groups can cause the disease to move in rapid jumps from village to city and back again.2
Framing a response: social theory
The Hewletts also raise an important conceptual issue central to the question of how communities adapt to the threat of Ebola. This is the issue of how beliefs and ideas relate to action.
As we will see below, international responders in 2013-15, led by the WHO, emphasized the importance of correcting wrong information. It was assumed that if communities had correct information on Ebola risks then appropriate actions would follow. This is a contentious assumption, because it takes sides in one of the most fundamental debates in social science - whether beliefs cause action, or actions cause belief.
The debate is perhaps best known through reactions to the early work of the German social philosopher Max Weber, who proposed that modern capitalism was significantly shaped by Calvinistic religious belief. The British economic historian R. H. Tawney tackled the same topic and period of history but arrived at a different conclusion, namely that ideas and economic practices co-evolved, with (in the last instance) material factors shaping the beliefs (including religious beliefs) of early capitalists.3
This has set the terms for a debate between materialists and idealists ever since. Social scientists of a materialist orientation sometimes build on the work of another German social philosopher, Karl Marx. Materialists who reject Marxist historical determinism prefer a variant of materialist thinking proposed by a third great nineteenth-century social philosopher, Emile Durkheim.4
Durkheim argued that belief is significantly shaped by social action. What we do, and how we do it as a group, including ritual action, shapes what we think we know about the world.
A choice made between these three perspectives significantly modifies how Ebola responders approach the key topic of rapid change of beliefs and practices regarding the sick and dead.
Broadly speaking, the Hewletts follow a Weberian route. They proffer the notion of ‘cultural models’ or ‘cultural scenarios’ to explain community Ebola response. These scenarios shape behaviour. The analysis is subtle. The Acholi people with whom the Hewletts worked in Uganda have a range of available mental templates, we are told, to explain disease and to guide response. They possess a specific cultural scenario for infectious disease, which includes notions of quarantine, and this was the one they applied to Ebola, thus allowing the Hewletts to explain how community action contributed to Ebola decline.
What is not explained is how the cultural scenarios came into existence, or why there should be (in the Acholi instance) three such scenarios, not two, or seven. With no theory of how the scenarios arise it is hard to see what responders would need to do to induce a helpful response were suitable cultural templates to be lacking.
Followers of Marx, we might suppose, would focus directly on the material factors shaping an Ebola epidemic. It is this kind of explanation that is engaged when attention is directed to the low level of investment in medical services in Upper West Africa as a driver of the disease.5
Marxist materialist explanation is challenged when the phenomenon of nosocomial infection is taken into consideration. This is the word applied to sicknesses that arise within medical treatment environments, and at times promoted by the treatment itself. Treatment of Ebola within a hospital environment is very risky both for other patients and for medical personnel. Having a better-funded treatment environment is not the answer. Treatment has to be specifically redesigned to separate Ebola patients and to protect responders at the earliest stage possible. This requires patients and responders to share ideas and beliefs appropriate to the kinds of behaviours that minimize infection risk. How this alignment of ideas, belief and behaviour is to be attained is a key issue for Ebola response.
In this book it will be suggested that it is insufficient to pump knowledge into the heads of affected populations, in the hope that this altered knowledge will guide behaviour change. The Durkheimian tradition in social theory argues that the relationship between mental and behavioural states is complex, and dependent not (alone) on individual cognition, but also on patterns of group action and interaction.
The compulsion to care for a sick person, or to take part in burying such a person after death, depends on drivers of group activity. One of the most fundamental drivers of behavioural commitment is the collective emotional state triggered and shaped by a ritual performance.
In effect, the Durkheimian position predicts that it will not be possible to prevent risky behaviour such as participation in burial services by outlining the medical risks. This type of action will change, in terms of its epidemiological effects, only when risky practices are replaced by ritually and emotionally meaningful equivalent safer practices. Devising these safer practices is work for community groups, not radio propagandists. A communication approach will make better sense when it reports the results of successful local ritual innovation. Support is needed, in the first instance, for affected community groups to devise their own new patterns of group interaction.
Thus what I seek to do in what follows (especially in Chapters 4 to 6) is to apply a Durkheimian perspective to the issue of local responses to Ebola. Beneficial change in community interaction dynamics around Ebola did in fact occur, but in a patchy manner. Local reorganizational efforts at times lacked the well-targeted external support that might have accelerated them and made their benefits more generally accessible. It will be argued that what external responders lacked most was a clear theory of local ritual interaction dynamics,6 and a support plan to enable communities to redirect their interaction dynamics in ways that would have been conducive to the rapid realignment of local beliefs and understandings with biosafety outcomes.
Ebola in Upper West Africa
The first recorded Ebola case in Upper West Africa occurred when a small boy fell sick and died in the village of Meliandou near the town of Guekedou (Gegedu), in the south-eastern part of the Republic of Guinea, during December 2013. The disease spread to members of the boy’s family and then to medical personnel, unaware that they were treating Ebola cases. The outbreak site was not in as isolated a region as in earlier outbreaks. Ebola cases soon began to spread down busy trading routes to Liberia and Sierra Leone as well.
The cumulative incidence of cases at 8 June 2015, when the epidemic was coming to an end, is shown in Table 1.2.
These figures reveal some variation between the three countries.
Judged in terms of numbers of fatalities Liberia was the worst-affected country. The figures need to be read with some caution, however. Table 1.2 shows a higher number of deaths for Liberia than confirmed cases. The rise of cases in Liberia was especially rapid, and for a time overwhelmed treatment and testing facilities. Clearly, data-gathering was a casualty, and we may never know, very exactly, what happened in Liberia in the earlier stages of the epidemic.
Table 1.2 Ebola cases in West Africa, 2013-15
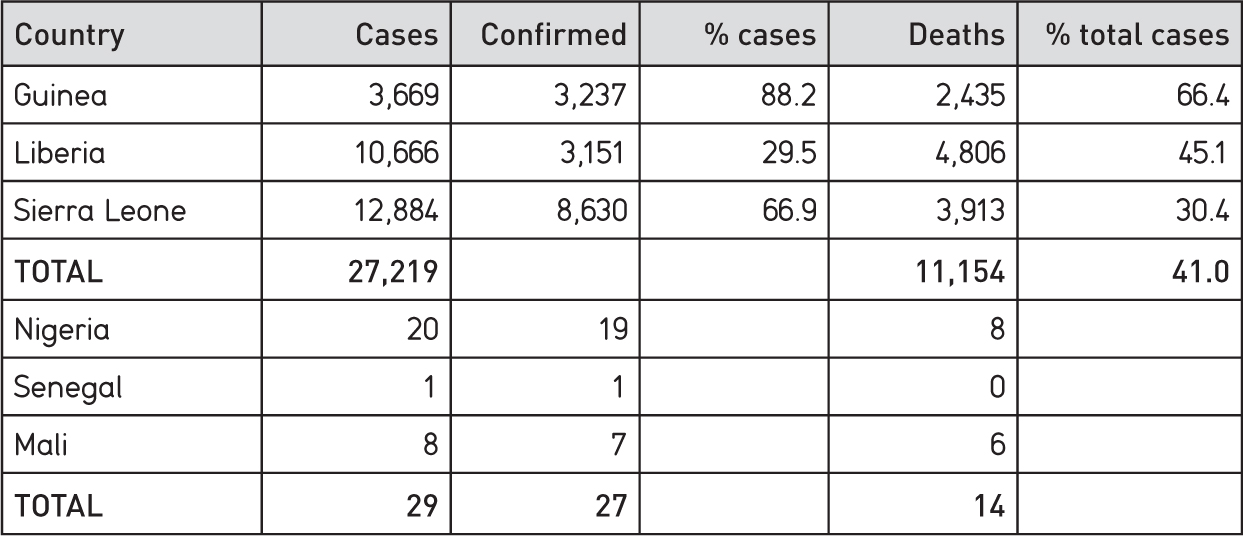
Source: CDC, 8 June 2015. Confirmed means laboratory tested. NB transmission was still active in Guinea and Sierra Leone on 8 June 2015, but had ended in other countries listed.
Sierra Leone recorded the largest number of total cases, but had the lowest death rate, well below the 50-90 per cent mortality widely cited for infection with the Zaire species of Ebola virus based on earlier epidemics. The ‘up to 90 per cent’ fatality figure was widely publicized by international media in the early days of the epidemic. It seems possible that this may have deterred some sick people from seeking help, particularly before local community care centres (CCCs) were introduced and family members could keep a daily eye on the fate of their loved ones.7
The epidemic began in Guinea, but took longest to control in this country (transmission ended in late November 2015). Several explanations have been offered for this persistence, including deep ethnic and political tensions between government and the people of the forest zone where the epidemic began.
These tensions may have fostered scepticism about the purpose of government-backed control measures. A second factor to be considered in the case of Guinea, however, is that here there were fewer cases, assessed against a total population of 12 million, relative to Liberia, 4.3 million, and Sierra Leone, 6 million. Experience of Ebola, and the learning that goes with it, was thus less intense in Guinea. In effect, virus transmission may have endured because it continued to find populations unfamiliar with the disease and measures needed to combat it.
The epidemic in Upper West Africa undoubtedly also had a head start because the initial response, both nationally and internationally, was slow. Although unexpected, the Guinea outbreak, once identified, was treated according to protocols applied in earlier isolated outbreaks. It was assumed the same procedures would work as well with the Guinea outbreak. Insufficient attention was paid to the intensive cross-border networking that catapulted the disease in the direction of adjacent, crowded, capital cities on the coast.
Established procedures worked well when outbreak areas were themselves highly isolated. No such distance barriers applied in the Upper West African cases. The forest region was within hours of the three large coastal cities by road, and international air travel connections rapidly took the virus farther.
This speed of spread caught Ebola experts by surprise. For instance, in Sierra Leone the first cases were confirmed in late May 2014, but by July there were cases in Freetown, the capital. An epidemic in city environments, with crowded slums, was uncharted terrain, and a degree of panic ensued.
Fortunately, this concern was misplaced. A study of Monrovia suggests that there is a statistically significant tendency for Ebola cases in the poorest urban districts to lead to more infective contacts than cases in wealthier neighbourhoods.8 But the difference is not very large, and urban slums did not prove to be the massive multipliers for an Ebola epidemic feared by some.9
A sense of panic overtaking the international community in mid-2014 was doubtless enhanced by widespread myths concerning the disease. Ebola has a certain sinister repute among those who model ‘doomwatch’ scenarios, not to mention those who watch Hollywood films.10
Both superpowers during the Cold War considered Ebola to be a potential biological weapon, and acquired genetic materials, perhaps mainly to try to figure out an antidote. The US Centers for Disease Control subsequently filed patents for some of the viral material it held, for vaccine development purposes.11
What was not known was whether Ebola would be a very good weapon of mass destruction. A virulent virus might all too readily backfire, and consume those who released it. Subsequent assessments suggested that the threat of Ebola would be more effective as a way of inducing panic than as a biological ‘bomb’.
This indeed proved to be true. The threat of ‘weaponized Ebola’, in the public mind, undoubtedly complicated response to the West African epidemic. In May 2015 a rumour circulated that two patients in a hospital in Mosul, Iraq had contracted Ebola.12 The fact that there was no known capacity in Iraq to test for Ebola did not dampen ensuing panic that Islamist rebels might be configuring a biological weapon.
So Guinea, Liberia and Sierra Leone found themselves fighting a war on two fronts. The first was against the first-ever epidemic of Ebola Virus Disease, but the second was a worldwide outbreak of media-inflamed Ebola Panic Disease.
A report by the World Health Organization, ‘Barriers to rapid containment of the Ebola outbreak’ (11 August 2014), cited fear as a major obstacle to rapid containment. The report noted that fear caused quarantined persons to flee, villagers to attack international responders, airlines to refuse to transport personal protective equipment, and courier services to refuse to transport properly and securely packaged patient samples to a WHO-approved laboratory.13
The British government, apparently mindful of possible adverse newspaper headlines as an election approached, halted direct flights to Sierra Leone and Liberia, against the advice of one of its own parliamentary committees.
The French government did something even stranger. Air France flights to Freetown and Monrovia were ended on account of Ebola risk, but not to Conakry. President Hollande explained it was a gesture of support for a francophone country. True, the spread of the virus in Guinea was never quite as rampant as in Liberia and Sierra Leone, but surely no one could have thought Ebola was less virulent on account of its linguistic orientation.
The three worst-affected countries, and their international partners, had to mobilize against the epidemic against this background noise.
A key step forward in the international response was when WHO declared Ebola in West Africa a Public Health Emergency of International Concern (PHEIC) on 8 August 2014. An international surge of response was then mobilized, even though it took several months before it was fully delivered.
A specialist UN agency, UNMEER, was founded to mobilize and coordinate this international response, and based in Ghana, a country without Ebola cases, but a choice perhaps dictated by the above-mentioned flight bans. French and Russian teams arrived to assist Guinea. British, Ugandan and other teams deployed to Sierra Leone. President Obama launched a major American initiative in Liberia, based around a large military mission.
Since Liberia was experiencing the most rapid rise in cases, the militarized American response began in August 2014. By contrast the surge in Sierra Leone did not fully get under way until December, when cases in that country peaked at 500+ a week, many of them in Freetown and its peri-urban fringe.
By early 2015 a trend towards decline, first apparent in Liberia in October 2014, was clear in all three countries. The extent to which this resulted from the international response, implementation of government controls, such as quarantine, local learning or epidemic ‘burn-out’, perhaps involving emergent natural immunities, will be discussed further in Chapters 2 and 3. Assigning probabilities to these different factors is worth attempting in order to consolidate understanding of prevention strategies relevant to any further emergence of Ebola on an epidemic scale.
Infection pathways
How did Ebola travel? Ebola has three main infection pathways. The first is termed zoonotic spillover. This pathway runs from forest animal host to human, often through consumption of ‘bushmeat’ (the name in West African English dialects for any kind of ‘game’). Monkeys and chimpanzees can become infected with Ebola, but develop symptoms and die. Some species of bats show evidence of carrying the virus but are apparently symptom free.
Bats are sometimes eaten extensively in, for example, parts of Ghana, Nigeria and Cameroon.14 Bat consumption is less common in and around the Upper West African forests.15 So the apparent spillover event in the Guinean epidemic remains puzzling. It seems unlikely a toddler (the index case) would have been a bat eater. It has been suggested he might have been playing in a bat-infected hollow tree, and became infected through bat droppings, but other stories circulate locally about possible earlier infections.16
On the other hand it soon became clear from work on genetic mutation rates of the virus in samples collected from patients in Sierra Leone that zoonotic spillover was not giving rise to new infections.17 The data from the mutational ‘clock’ were consistent with the interpretation that all further infection had been human-to-human.
Virologists traced three distinct lineages of the Makona variant of the Ebola virus in Sierra Leone, based on blood samples from patients with confirmed cases. The third lineage (SL3) emerged in mid-June 2014, and a recent study found that 97 per cent of its samples carried this variant of the genome.18 These results ‘link all Sierra Leonean … cases to the initial introduction of [the virus] into Sierra Leone, and … provide further evidence that all … cases during this outbreak arose from human-to-human transmission rather than from further zoonotic introductions from an unknown … reservoir [of the virus]’.19 Furthermore (the same report adds), the genetic similarity of these viruses ‘suggests that importation from other countries was minimal’, though the authors do not rule this out entirely.20
The other two major pathways for infection involve human-to-human contact.
One is nosocomial (infection as a result of treatment in a hospital or clinic). Any medical facility not up to the highest level of biosecurity standards can cross-infect other patients with Ebola. This biosecurity capacity is found in only a very few hospitals in the Western world, so it should not be assumed, as some appear to have done, that nosocomial transmission makes Ebola a ‘disease of poverty’, spreading in run-down medical facilities starved of staff and funds. Ordinary hygiene cannot protect against Ebola. Specific practices must be implemented, based on a thorough understanding of infection risk.
The second ‘person-to-person’ infection pathway is through family care. Ebola is a ‘nasty disease’21 because it punishes carers for not abandoning their loved ones. The main risks, from virus load in body fluids, are encountered in the ‘wet’ phase of the sickness, and in preparing the corpse for burial. Without behaviour modification a patient will infect household carers, and the group involved in washing the dead body. Touching the body to say farewell, or handing over grave cloth as a keepsake, may further increase the numbers of post-mortem infections.
Some large-scale funerals of prominent persons led to the infection of scores of mourners. The initial fourteen confirmed cases in Sierra Leone, for instance, were all epidemiologically linked to the funeral of a traditional healer.22Agencies involved in the Ebola response in Sierra Leone later ‘claimed that 70 per cent of new infections stemmed from funeral rituals’.23
Requirements for ending an Ebola epidemic
Six things must be in place to halt spread of infection.
1.Ebola victims must be identified (through reporting cases to a special telephone helpline, and rapid diagnostic testing).
2.Ebola-positive cases must then be isolated, preferably in a purpose-built treatment facility, with safe drainage and waste disposal; carers must have disposable protective gear (PPE), and stringent hygiene must be followed.
3.Patients need to be transported in special ambulances by trained teams, using PPE, to reduce risks of cross-infection.
4.There is a need to consider quarantine. Quarantine is controversial, but is still probably a necessary step. Households from which Ebola patients have been extracted were in many cases quarantined for 21 days and supplied with basic provisions. This sometimes proved counter-productive, in encouraging people to abscond. Local efforts at quarantine may be more relevant to disease control, being focused on the need to restrict movement into communities of strangers with unknown health status.
5.All close contacts of the sick person need to be traced and visited every day for twenty-one days to check for signs of sickness. This is a huge logistic undertaking. In Sierra Leone, for example, as of 9 June 2015, a total of 96,768 contacts had been followed up daily, each for twenty-one days (2,033 contacts did not finish the twenty-one days owing to death or other factors).
6.All deaths must be reported and ‘safe burial’ procedures applied. A trained team arrives to prepare the body, and wraps it in two body bags, before the funeral takes place. Swabs are taken to establish whether the death was an Ebola case. The family is allowed to witness from a distance, but the interment itself is carried out by the team. It should perhaps be emphasized that this applies to all deaths, whether from Ebola or not. All bereaved families are affected by Ebola, and not only those in which a death from Ebola has occurred.
All six areas just mentioned pose severe challenges of social acceptance. This is where the issues of social theory outlined above become relevant. The predominant approach in the response to the Upper West African Ebola epidemic in 2013/15 was to assume that ideas drive behaviour. To change ideas it was assumed that better information was required. Social mobilization was taken to mean a focus on messages.
Some commentators canvassed alternatives based on a materialist approach. According to this viewpoint the spread of Ebola was related to the inadequacy of medical provision. Better provision would have reduced the risks of an epidemic. But faced with the challenge of rapidly rising numbers of victims this was a counsel of perfection.
It might be better to approach Ebola prevention through an analysis of how collective action (including ritual action) supports belief. In particular, it seems important to analyse collective action in terms of body technique. How exactly was patient care delivered, and how exactly were bodies buried? This in turn might offer some insight into community capacity for changing relevant areas of embodied performance. The states of belief sustained by modes of collective action were in a sense of little account. What mattered was the extent to which communities at risk of Ebola were capable of recognizing the risks posed by their existing embodied practices and to elaborate safer techniques. Messaging was less important than scope for practice.
The following chapters thus focus on two central concerns: the way in which existing embodied techniques influence biosafety in regard to Ebola infection pathways, and the scope for endogenous change in such techniques. The case is made for a people’s science of Ebola control generated through rapid group learning concerning behavioural modification. Consolidating such newly acquired capacities in communities exposed to Ebola, so that they remain available over the longer term, and disseminating these skills more widely to communities not yet affected, is a suggested prerequisite for post-Ebola preparedness throughout Africa.